Abstract
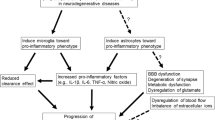
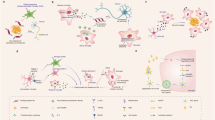
One of the most striking hallmarks shared by various neurodegenerative diseases, including Parkinson’s disease, Alzheimer’s disease (AD), and amyotrophic lateral sclerosis, is microglia-mediated neuroinflammation. Increasing evidence indicates that microglial activation in the central nervous system is heterogeneous, which can be categorized into two opposite types: M1 phenotype and M2 phenotype. Depending on the phenotypes activated, microglia can produce either cytotoxic or neuroprotective effects. In this review, we focus on the potential role of M1 and M2 microglia and the dynamic changes of M1/M2 phenotypes that are critically associated with the neurodegenerative diseases. Generally, M1 microglia predominate at the injury site at the end stage of disease, when the immunoresolution and repair process of M2 microglia are dampened. This phenotype transformation is very complicated in AD due to the phagocytosis of regionally distributed β-amyloid (Aβ) plaque and tangles that are released into the extracellular space. The endogenous stimuli including aggregated α-synuclein, mutated superoxide dismutase, Aβ, and tau oligomers exist in the milieu that may persistently activate M1 pro-inflammatory responses and finally lead to irreversible neuron loss. The changes of microglial phenotypes depend on the disease stages and severity; mastering the stage-specific switching of M1/M2 phenotypes within appropriate time windows may provide better therapeutic benefit.



Similar content being viewed by others
Abbreviations
- AD:
-
Alzheimer’s disease
- ALS:
-
Amyotrophic lateral sclerosis
- APP:
-
Amyloid precursor protein
- Arg1:
-
Arginase 1
- Aβ:
-
β-Amyloid
- BDNF:
-
Brain-derived neurotrophic factor
- CD206:
-
Mannose receptor
- Chi3l3:
-
Chitinase-3-Like-3
- CNS:
-
Central nervous system
- DA:
-
Dopaminergic
- ECM:
-
Extracellular matrix
- FIZZ1:
-
Found in inflammatory zone 1
- IFN-γ:
-
Interferon-γ
- IGF-I:
-
Insulin-like growth factor 1
- IL:
-
Interleukin
- iNOS:
-
Induced nitric oxide synthase
- LBs:
-
Lewy bodies
- LPS:
-
Lipopolysaccharide
- MHC:
-
Major histocompatibility complex
- MPTP:
-
1-Methyl-4-phenyl-1,2,3,6-tetrahydropyridine
- mSOD1:
-
Mutated superoxide dismutase
- NO:
-
Nitric oxide
- PD:
-
Parkinson’s disease
- PET:
-
Positron emission tomography
- PS1:
-
Presenilin-1
- RELM:
-
Resistin-like molecules
- ROS:
-
Reactive oxygen species
- SN:
-
Substantia nigra
- SRA:
-
Scavenger receptors
- TAM:
-
Tumor-associated macrophages
- TDP-43:
-
TAR DNA-binding protein 43
- TGF-β:
-
Transforming growth factor-β
- TLRs:
-
Toll-like receptors
- TNF-α:
-
Tumor necrosis factor-α
- TTBK:
-
Tau-tubulin kinase
References
Gao HM, Hong JS (2008) Why neurodegenerative diseases are progressive: uncontrolled inflammation drives disease progression. Trends Immunol 29(8):357–365
Glass CK, Saijo K, Winner B, Marchetto MC, Gage FH (2010) Mechanisms underlying inflammation in neurodegeneration. Cell 140(6):918–934
Minghetti L (2005) Role of inflammation in neurodegenerative diseases. Curr Opin Neurol 18(3):315–321
Block ML, Hong JS (2005) Microglial and inflammation-mediated neurodegeneration: multiple triggers with a common mechanism. Prog Neurobiol 76(2):77–98
Ouchi Y, Yoshikawa E, Sekine Y, Futatsubashi M, Kanno T, Ogusu T et al (2005) Microglial activation and dopamine terminal loss in early Parkinson’s disease. Ann Neurol 57(2):168–175
Block ML, Zecca L, Hong JS (2007) Microglia-mediated neurotoxicity: uncovering the molecular mechanisms. Nat Rev Neurosci 8(1):57–69
Gerhard A, Pavese N, Hotton G, Turkheimer F, Es M, Hammers A et al (2006) In vivo imaging of microglial activation with [11C](R)-PK11195 PET in idiopathic Parkinson’s disease. Neurobiol Dis 21(2):404–412
Hanisch UK, Kettenmann H (2007) Microglia: active sensor and versatile effector cells in the normal and pathologic brain. Nat Neurosci 10(11):1387–1394
Tang Y, Le W (2014) “Good” and “bad” microglia in Parkinson’s disease: an understanding of homeostatic mechanisms in immunomodulation. In: Thomas M (ed) Inflammation in Parkinson’s disease. Springer, New York, pp 105–126
Glezer I, Simard AR, Rivest S (2007) Neuroprotective role of the innate immune system by microglia. Neuroscience 147(4):867–883
Simard AR, Soulet D, Gowing G, Julien JP, Rivest S (2006) Bone marrow-derived microglia play a critical role in restricting senile plaque formation in Alzheimer’s disease. Neuron 49(4):489–502
Ding YM, Jaumotte JD, Signore AP, Zigmond MJ (2004) Effects of 6-hydroxydopamine on primary cultures of substantia nigra: specific damage to dopamine neurons and the impact of glial cell line-derived neurotrophic factor. J Neurochem 89(3):776–787
Lumeng CN, Bodzin JL, Saltiel AR (2007) Obesity induces a phenotypic switch in adipose tissue macrophage polarization. J Clin Invest 117(1):175–184
Lumeng CN, Deyoung SM, Bodzin JL, Saltiel AR (2007) Increased inflammatory properties of adipose tissue macrophages recruited during diet-induced obesity. Diabetes 56(1):16–23
Odegaard JI, Ricardo-Gonzalez RR, Goforth MH, Morel CR, Subramanian V, Mukundan L et al (2007) Macrophage-specific PPARgamma controls alternative activation and improves insulin resistance. Nature 447(7148):1116–1120
Hevener AL, Olefsky JM, Reichart D, Nguyen MT, Bandyopadyhay G, Leung HY et al (2007) Macrophage PPAR gamma is required for normal skeletal muscle and hepatic insulin sensitivity and full antidiabetic effects of thiazolidinediones. J Clin Invest 117(6):1658–1669
Kigerl KA, Gensel JC, Ankeny DP, Alexander JK, Donnelly DJ, Popovich PG (2009) Identification of two distinct macrophage subsets with divergent effects causing either neurotoxicity or regeneration in the injured mouse spinal cord. J Neurosci 29(43):13435–13444
Colton C, Wilcock DM (2010) Assessing activation states in microglia. CNS Neurol Disord Drug Targets 9(2):174–191
Colton CA (2009) Heterogeneity of microglial activation in the innate immune response in the brain. J Neuroimmune Pharmacol 4(4):399–418
Le W, Rowe D, Xie W, Ortiz I, He Y, Appel SH (2001) Microglial activation and dopaminergic cell injury: an in vitro model relevant to Parkinson’s disease. J Neurosci 21(21):8447–8455
Li R, Huang YG, Fang D, Le WD (2004) (−)-Epigallocatechin gallate inhibits lipopolysaccharide-induced microglial activation and protects against inflammation-mediated dopaminergic neuronal injury. J Neurosci Res 78(5):723–731
Ponomarev ED, Maresz K, Tan Y, Dittel BN (2007) CNS-derived interleukin-4 is essential for the regulation of autoimmune inflammation and induces a state of alternative activation in microglial cells. J Neurosci 27(40):10714–10721
Sawada M, Suzumura A, Hosoya H, Marunouchi T, Nagatsu T (1999) Interleukin-10 inhibits both production of cytokines and expression of cytokine receptors in microglia. J Neurochem 72(4):1466–1471
Gao HM, Liu B, Zhang WQ, Hong JS (2003) Critical role of microglial NADPH oxidase-derived free radicals in the in vitro MPTP model of Parkinson’s disease. FASEB J 17(11):1954−+
Butovsky O, Talpalar AE, Ben-Yaakov K, Schwartz M (2005) Activation of microglia by aggregated beta-amyloid or lipopolysaccharide impairs MHC-II expression and renders them cytotoxic whereas IFN-gamma and IL-4 render them protective. Mol Cell Neurosci 29(3):381–393
Zhou XL, Spittau B, Krieglstein K (2012) TGF beta signalling plays an important role in IL4-induced alternative activation of microglia. J Neuroinflamm 9:210–223
Ledeboer A, Breve JJ, Poole S, Tilders FJ, Van Dam AM (2000) Interleukin-10, interleukin-4, and transforming growth factor-beta differentially regulate lipopolysaccharide-induced production of pro-inflammatory cytokines and nitric oxide in co-cultures of rat astroglial and microglial cells. Glia 30(2):134–142
Zhao WH, Xie WJ, Xiao Q, Beers DR, Appel SH (2006) Protective effects of an anti-inflammatory cytokine, interleukin-4, on motoneuron toxicity induced by activated microglia. J Neurochem 99(4):1176–1187
Park KW, Lee DY, Joe EH, Kim SU, Jin BK (2005) Neuroprotective role of microglia expressing interleukin-4. J Neurosci Res 81(3):397–402
Boche D, Cunningham C, Docagne F, Scott H, Perry VH (2006) TGFbeta1 regulates the inflammatory response during chronic neurodegeneration. Neurobiol Dis 22(3):638–650
Boche D, Cunningham C, Gauldie J, Perry VH (2003) Transforming growth factor-beta 1-mediated neuroprotection against excitotoxic injury in vivo. J Cereb Blood Flow Metab 23(10):1174–1182
Bogdan C, Vodovotz Y, Nathan C (1991) Macrophage deactivation by interleukin 10. J Exp Med 174(6):1549–1555
Buechler C, Ritter M, Orso E, Langmann T, Klucken J, Schmitz G (2000) Regulation of scavenger receptor CD163 expression in human monocytes and macrophages by pro- and antiinflammatory stimuli. J Leukoc Biol 67(1):97–103
Fabriek BO, Dijkstra CD, van den Berg TK (2005) The macrophage scavenger receptor CD163. Immunobiology 210(2–4):153–160
Suh HS, Zhao ML, Derico L, Choi N, Lee SC (2013) Insulin-like growth factor 1 and 2 (IGF1, IGF2) expression in human microglia: differential regulation by inflammatory mediators. J Neuroinflamm 10:37
Liu Y, Hao W, Letiembre M, Walter S, Kulanga M, Neumann H et al (2006) Suppression of microglial inflammatory activity by myelin phagocytosis: role of p47-PHOX-mediated generation of reactive oxygen species. J Neurosci 26(50):12904–12913
Gordon S (2003) Alternative activation of macrophages. Nat Rev Immunol 3(1):23–35
Morris SM (2004) Recent advances in arginine metabolism. Curr Opin Clin Nutr 7(1):45–51
Bronte V, Zanovello P (2005) Regulation of immune responses by L-arginine metabolism. Nat Rev Immunol 5(8):641–654
Busch SA, Silver J (2007) The role of extracellular matrix in CNS regeneration. Curr Opin Neurobiol 17(1):120–127
Wu GY, Bazer FW, Burghardt RC, Johnson GA, Kim SW, Knabe DA et al (2011) Proline and hydroxyproline metabolism: implications for animal and human nutrition. Amino Acids 40(4):1053–1063
Jenkins CL, Bretscher LE, Guzei IA, Raines RT (2003) Effect of 3-hydroxyproline residues on collagen stability. J Am Chem Soc 125(21):6422–6427
Thomas T, Thomas TJ (2001) Polyamines in cell growth and cell death: molecular mechanisms and therapeutic applications. Cell Mol Life Sci 58(2):244–258
Williams K (1997) Interactions of polyamines with ion channels. Biochem J 325:289–297
Tang Y, Li T, Li J, Yang J, Liu H, Zhang XJ et al (2014) Jmjd3 is essential for the epigenetic modulation of microglia phenotypes in the immune pathogenesis of Parkinson’s disease. Cell Death Differ 21(3):369–380
Colton CA, Mott RT, Sharpe H, Xu Q, Van Nostrand WE, Vitek MP (2006) Expression profiles for macrophage alternative activation genes in AD and in mouse models of AD. J Neuroinflamm 3:27
Nair MG, Du YR, Perrigoue JG, Zaph C, Taylor JJ, Goldschmidt M et al (2009) Alternatively activated macrophage-derived RELM-alpha is a negative regulator of type 2 inflammation in the lung (vol 206, pg 397, 2009). J Exp Med 206(5):1201
Holcomb IN, Kabakoff RC, Chan B, Baker TW, Gurney A, Henzel W et al (2000) FIZZ1, a novel cysteine-rich secreted protein associated with pulmonary inflammation, defines a new gene family. EMBO J 19(15):4046–4055
Munitz A, Seidu L, Cole ET, Ahrens R, Hogan SP, Rothenberg ME (2009) Resistin-like molecule a decreases glucose tolerance during intestinal inflammation. J Immunol 182(4):2357–2363
Munitz A, Waddell A, Seidu L, Ahrens R, Hogan SP, Rothenberg ME (2008) Resistin-like molecule alpha enhances myeloid cell activation and promotes colitis. J Allergy Clin Immun 122(6):1200–1207
Chang NCA, Hung SI, Hwa KY, Kato I, Chen JE, Liu CH et al (2001) A macrophage protein, Ym1, transiently expressed during inflammation is a novel mammalian lectin. J Biol Chem 276(20):17497–17506
Hung SL, Chang AC, Kato I, Chang NCA (2002) Transient expression of Ym1, a heparin-binding lectin, during developmental hematopoiesis and inflammation. J Leukocyte Biol 72(1):72–82
Recklies AD, White C, Ling H (2002) The chitinase 3-like protein human cartilage glycoprotein 39 (HC-gp39) stimulates proliferation of human connective-tissue cells and activates both extracellular signal-regulated kinase- and protein kinase beta-mediated signalling pathways. Biochem J 365:119–126
Raes G, De Baetselier P, Noel W, Beschin A, Brombacher F, Hassanzadeh G (2002) Differential expression of FIZZ1 and Ym1 in alternatively versus classically activated macrophages. J Leukocyte Biol 71(4):597–602
Welch JS, Escoubet-Lozach L, Sykes DB, Liddiard K, Greaves DR, Glass CK (2002) T(H)2 cytokines and allergic challenge induce YM1 expression in macrophages by a STAT6-dependent mechanism. J Biol Chem 277(45):42821–42829
Lee E, Yook J, Haa K, Chang HW (2005) Induction of Ym1/2 in mouse bone marrow-derived mast cells by IL-4 and identification of Ym1/2 in connective tissue type-like mast cells derived from bone marrow cells cultured with IL-4 and stem cell factor. Immunol Cell Biol 83(5):468–474
Choi SH, Aid S, Kim HW, Jackson SH, Bosetti F (2012) Inhibition of NADPH oxidase promotes alternative and anti-inflammatory microglial activation during neuroinflammation. J Neurochem 120(2):292–301
Taylor PR, Gordon S, Martinez-Pomares L (2005) The mannose receptor: linking homeostasis and immunity through sugar recognition. Trends Immunol 26(2):104–110
Stahl PD, Ezekowitz RAB (1998) The mannose receptor is a pattern recognition receptor involved in host defense. Curr Opin Immunol 10(1):50–55
Lee SJ, Evers S, Roeder D, Parlow AF, Risteli J, Risteli L et al (2002) Mannose receptor-mediated regulation of serum glycoprotein homeostasis. Science 295(5561):1898–1901
Chieppa M, Bianchi G, Doni A, Del Prete A, Sironi M, Laskarin G et al (2003) Cross-linking of the mannose receptor on monocyte-derived dendritic cells activates an anti-inflammatory immunosuppressive program. J Immunol 171(9):4552–4560
Kerrigan AM, Brown GD (2009) C-type lectins and phagocytosis. Immunobiology 214(7):562–575
Kigerl KA, Gensel JC, Ankeny DP, Alexander JK, Donnelly DJ, Popovich PG (2009) Identification of two distinct macrophage subsets with divergent effects causing either neurotoxicity or regeneration in the injured mouse spinal cord. J Neurosci 29(43):13435–13444
Madsen DH, Leonard D, Masedunskas A, Moyer A, Jurgensen HJ, Peters DE et al (2013) M2-like macrophages are responsible for collagen degradation through a mannose receptor-mediated pathway. J Cell Biol 202(6):951–966
Dauer W, Przedborski S (2003) Parkinson’s disease: mechanisms and models. Neuron 39(6):889–909
Braak H, Del Tredici K, Rub U, de Vos RA, Jansen Steur EN, Braak E (2003) Staging of brain pathology related to sporadic Parkinson’s disease. Neurobiol Aging 24(2):197–211
Lashuel HA, Overk CR, Oueslati A, Masliah E (2013) The many faces of alpha-synuclein: from structure and toxicity to therapeutic target. Nat Rev Neurosci 14(1):38–48
Polymeropoulos MH, Lavedan C, Leroy E, Ide SE, Dehejia A, Dutra A et al (1997) Mutation in the alpha-synuclein gene identified in families with Parkinson’s disease. Science 276(5321):2045–2047
Klein C, Westenberger A (2012) Genetics of Parkinson’s disease. CSH Perspect Med 2(1)
Sanchez-Guajardo V, Barnum CJ, Tansey MG, Romero-Ramos M (2013) Neuroimmunological processes in Parkinson’s disease and their relation to alpha-synuclein: microglia as the referee between neuronal processes and peripheral immunity. ASN Neurol 5(2):113–139
Zhang W, Wang T, Pei Z, Miller DS, Wu X, Block ML et al (2005) Aggregated alpha-synuclein activates microglia: a process leading to disease progression in Parkinson’s disease. FASEB J 19(6):533–542
Lee EJ, Woo MS, Moon PG, Baek MC, Choi IY, Kim WK et al (2010) Alpha-synuclein activates microglia by inducing the expressions of matrix metalloproteinases and the subsequent activation of protease-activated receptor-1. J Immunol 185(1):615–623
Zhang W, Dallas S, Zhang D, Guo JP, Pang H, Wilson B et al (2007) Microglial PHOX and Mac-1 are essential to the enhanced dopaminergic neurodegeneration elicited by A30P and A53T mutant alpha-synuclein. Glia 55(11):1178–1188
Reynolds AD, Kadiu I, Garg SK, Glanzer JG, Nordgren T, Ciborowski P et al (2008) Nitrated alpha-synuclein and microglial neuroregulatory activities. J Neuroimmune Pharmacol 3(2):59–74
Gao HM, Kotzbauer PT, Uryu K, Leight S, Trojanowski JQ, Lee VM (2008) Neuroinflammation and oxidation/nitration of alpha-synuclein linked to dopaminergic neurodegeneration. J Neurosci 28(30):7687–7698
Rojanathammanee L, Murphy EJ, Combs CK (2011) Expression of mutant alpha-synuclein modulates microglial phenotype in vitro. J Neuroinflamm 8:44
Austin SA, Floden AM, Murphy EJ, Combs CK (2006) Alpha-synuclein expression modulates microglial activation phenotype. J Neurosci 26(41):10558–10563
Porras G, Li Q, Bezard E (2012) Modeling Parkinson’s disease in primates: the MPTP model. CSH Perspect Med 2(3)
Wu DC, Teismann P, Tieu K, Vila M, Jackson-Lewis V, Ischiropoulos H et al (2003) NADPH oxidase mediates oxidative stress in the 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine model of Parkinson’s disease. Proc Natl Acad Sci U S A 100(10):6145–6150
Zhang W, Wang T, Qin L, Gao HM, Wilson B, Ali SF et al (2004) Neuroprotective effect of dextromethorphan in the MPTP Parkinson’s disease model: role of NADPH oxidase. FASEB J 18(3):589–591
Liberatore GT, Jackson-Lewis V, Vukosavic S, Mandir AS, Vila M, McAuliffe WG et al (1999) Inducible nitric oxide synthase stimulates dopaminergic neurodegeneration in the MPTP model of Parkinson disease. Nat Med 5(12):1403–1409
Gao HM, Jiang J, Wilson B, Zhang W, Hong JS, Liu B (2002) Microglial activation-mediated delayed and progressive degeneration of rat nigral dopaminergic neurons: relevance to Parkinson’s disease. J Neurochem 81(6):1285–1297
Theodore S, Cao S, McLean PJ, Standaert DG (2008) Targeted overexpression of human alpha-synuclein triggers microglial activation and an adaptive immune response in a mouse model of Parkinson disease. J Neuropathol Exp Neurol 67(12):1149–1158
Zhang H, Li Y, Yu J, Guo M, Meng J, Liu C et al (2013) Rho kinase inhibitor fasudil regulates microglia polarization and function. Neuroimmunomodulat 20(6):313–322
Mattson MP (2004) Pathways towards and away from Alzheimer’s disease. Nature 430(7000):631–639
Citron M, Oltersdorf T, Haass C, Mcconlogue L, Hung AY, Seubert P et al (1992) Mutation of the beta-amyloid precursor protein in familial Alzheimer’s-disease increases beta-protein production. Nature 360(6405):672–674
Meyer-Luehmann M, Spires-Jones TL, Prada C, Garcia-Alloza M, de Calignon A, Rozkalne A et al (2008) Rapid appearance and local toxicity of amyloid-beta plaques in a mouse model of Alzheimer’s disease. Nature 451(7179):720–724
Maezawa I, Zimin PI, Wulff H, Jin LW (2011) Amyloid-beta protein oligomer at low nanomolar concentrations activates microglia and induces microglial neurotoxicity. J Biol Chem 286(5):3693–3706
Walker DG, Link J, Lue LF, Dalsing-Hernandez JE, Boyes BE (2006) Gene expression changes by amyloid beta peptide-stimulated human postmortem brain microglia identify activation of multiple inflammatory processes. J Leukoc Biol 79(3):596–610
Takata K, Kitamura Y, Saeki M, Terada M, Kagitani S, Kitamura R et al (2010) Galantamine-induced amyloid-{beta} clearance mediated via stimulation of microglial nicotinic acetylcholine receptors. J Biol Chem 285(51):40180–40191
Mawuenyega KG, Sigurdson W, Ovod V, Munsell L, Kasten T, Morris JC et al (2010) Decreased clearance of CNS beta-amyloid in Alzheimer’s disease. Science 330(6012):1774
Jimenez S, Baglietto-Vargas D, Caballero C, Moreno-Gonzalez I, Torres M, Sanchez-Varo R et al (2008) Inflammatory response in the hippocampus of PS1M146L/APP751SL mouse model of Alzheimer’s disease: age-dependent switch in the microglial phenotype from alternative to classic. J Neurosci 28(45):11650–11661
Koenigsknecht-Talboo J, Landreth GE (2005) Microglial phagocytosis induced by fibrillar beta-amyloid and IgGs are differentially regulated by proinflammatory cytokines. J Neurosci 25(36):8240–8249
Michelucci A, Heurtaux T, Grandbarbe L, Morga E, Heuschling P (2009) Characterization of the microglial phenotype under specific pro-inflammatory and anti-inflammatory conditions: effects of oligomeric and fibrillar amyloid-beta. J Neuroimmunol 210(1–2):3–12
Butovsky O, Koronyo-Hamaoui M, Kunis G, Ophir E, Landa G, Cohen H et al (2006) Glatiramer acetate fights against Alzheimer’s disease by inducing dendritic-like microglia expressing insulin-like growth factor 1. Proc Natl Acad Sci U S A 103(31):11784–11789
Lyons A, Griffin RJ, Costelloe CE, Clarke RM, Lynch MA (2007) IL-4 attenuates the neuroinflammation induced by amyloid-beta in vivo and in vitro. J Neurochem 101(3):771–781
Iribarren P, Chen K, Hu J, Zhang X, Gong W, Wang JM (2005) IL-4 inhibits the expression of mouse formyl peptide receptor 2, a receptor for amyloid beta1-42, in TNF-alpha-activated microglia. J Immunol 175(9):6100–6106
Tichauer JE, von Bernhardi R (2012) Transforming growth factor-beta stimulates beta amyloid uptake by microglia through Smad3-dependent mechanisms. J Neurosci Res 90(10):1970–1980
Nathan C, Calingasan N, Nezezon J, Ding A, Lucia MS, La Perle K et al (2005) Protection from Alzheimer’s-like disease in the mouse by genetic ablation of inducible nitric oxide synthase. J Exp Med 202(9):1163–1169
Herber DL, Mercer M, Roth LM, Symmonds K, Maloney J, Wilson N et al (2007) Microglial activation is required for Abeta clearance after intracranial injection of lipopolysaccharide in APP transgenic mice. J Neuroimmune Pharmacol 2(2):222–231
Herber DL, Roth LM, Wilson D, Wilson N, Mason JE, Morgan D et al (2004) Time-dependent reduction in Abeta levels after intracranial LPS administration in APP transgenic mice. Exp Neurol 190(1):245–253
DiCarlo G, Wilcock D, Henderson D, Gordon M, Morgan D (2001) Intrahippocampal LPS injections reduce Abeta load in APP + PS1 transgenic mice. Neurobiol Aging 22(6):1007–1012
Wilcock DM, Lewis MR, Van Nostrand WE, Davis J, Previti ML, Gharkholonarehe N et al (2008) Progression of amyloid pathology to Alzheimer’s disease pathology in an amyloid precursor protein transgenic mouse model by removal of nitric oxide synthase 2. J Neurosci 28(7):1537–1545
Chakrabarty P, Ceballos-Diaz C, Beccard A, Janus C, Dickson D, Golde TE et al (2010) IFN-gamma promotes complement expression and attenuates amyloid plaque deposition in amyloid beta precursor protein transgenic mice. J Immunol 184(9):5333–5343
Chakrabarty P, Jansen-West K, Beccard A, Ceballos-Diaz C, Levites Y, Verbeeck C et al (2010) Massive gliosis induced by interleukin-6 suppresses Abeta deposition in vivo: evidence against inflammation as a driving force for amyloid deposition. FASEB J 24(2):548–559
Chakrabarty P, Herring A, Ceballos-Diaz C, Das P, Golde TE (2011) Hippocampal expression of murine TNFalpha results in attenuation of amyloid deposition in vivo. Mol Neurodegener 6:16
Kawahara K, Suenobu M, Yoshida A, Koga K, Hyodo A, Ohtsuka H et al (2012) Intracerebral microinjection of interleukin-4/interleukin-13 reduces beta-amyloid accumulation in the ipsilateral side and improves cognitive deficits in young amyloid precursor protein 23 mice. Neuroscience 207:243–260
Kiyota T, Okuyama S, Swan RJ, Jacobsen MT, Gendelman HE, Ikezu T (2010) CNS expression of anti-inflammatory cytokine interleukin-4 attenuates Alzheimer’s disease-like pathogenesis in APP + PS1 bigenic mice. FASEB J 24(8):3093–3102
Kiyota T, Ingraham KL, Swan RJ, Jacobsen MT, Andrews SJ, Ikezu T (2012) AAV serotype 2/1-mediated gene delivery of anti-inflammatory interleukin-10 enhances neurogenesis and cognitive function in APP + PS1 mice. Gene Ther 19(7):724–733
Chakrabarty P, Tianbai L, Herring A, Ceballos-Diaz C, Das P, Golde TE (2012) Hippocampal expression of murine IL-4 results in exacerbation of amyloid deposition. Mol Neurodegener 7:36
Colangelo V, Schurr J, Ball MJ, Pelaez RP, Bazan NG, Lukiw WJ (2002) Gene expression profiling of 12633 genes in Alzheimer hippocampal CA1: transcription and neurotrophic factor down-regulation and up-regulation of apoptotic and pro-inflammatory signaling. J Neurosci Res 70(3):462–473
Blalock EM, Geddes JW, Chen KC, Porter NM, Markesbery WR, Landfield PW (2004) Incipient Alzheimer’s disease: microarray correlation analyses reveal major transcriptional and tumor suppressor responses. Proc Natl Acad Sci U S A 101(7):2173–2178
Serrano-Pozo A, Mielke ML, Gomez-Isla T, Betensky RA, Growdon JH, Frosch MP et al (2011) Reactive glia not only associates with plaques but also parallels tangles in Alzheimer’s disease. Am J Pathol 179(3):1373–1384
Ikeda M, Shoji M, Kawarai T, Kawarabayashi T, Matsubara E, Murakami T et al (2005) Accumulation of filamentous tau in the cerebral cortex of human tau R406W transgenic mice. Am J Pathol 166(2):521–531
Zilka N, Stozicka Z, Kovac A, Pilipcinec E, Bugos O, Novak M (2009) Human misfolded truncated tau protein promotes activation of microglia and leukocyte infiltration in the transgenic rat model of tauopathy. J Neuroimmunol 209(1–2):16–25
Kovac A, Zilka N, Kazmerova Z, Cente M, Zilkova M, Novak M (2011) Misfolded truncated protein tau induces innate immune response via MAPK pathway. J Immunol 187(5):2732–2739
Sasaki A, Kawarabayashi T, Murakami T, Matsubara E, Ikeda M, Hagiwara H et al (2008) Microglial activation in brain lesions with tau deposits: comparison of human tauopathies and tau transgenic mice TgTauP301L. Brain Res 1214:159–168
Morales I, Jimenez JM, Mancilla M, Maccioni RB (2013) Tau oligomers and fibrils induce activation of microglial cells. J Alzheimers Dis 37(4):849–856
Zilka N, Kazmerova Z, Jadhav S, Neradil P, Madari A, Obetkova D et al (2012) Who fans the flames of Alzheimer’s disease brains? Misfolded tau on the crossroad of neurodegenerative and inflammatory pathways. J Neuroinflamm 9:47
Bhaskar K, Konerth M, Kokiko-Cochran ON, Cardona A, Ransohoff RM, Lamb BT (2010) Regulation of tau pathology by the microglial fractalkine receptor. Neuron 68(1):19–31
Wes PD, Easton A, Corradi J, Barten DM, Devidze N, DeCarr LB et al (2014) Tau overexpression impacts a neuroinflammation gene expression network perturbed in Alzheimer’s disease. PLoS One 9(8):e106050
Yoshiyama Y, Higuchi M, Zhang B, Huang SM, Iwata N, Saido TC et al (2007) Synapse loss and microglial activation precede tangles in a P301S tauopathy mouse model. Neuron 53(3):337–351
Lewis J, Dickson DW, Lin WL, Chisholm L, Corral A, Jones G et al (2001) Enhanced neurofibrillary degeneration in transgenic mice expressing mutant tau and APP. Science 293(5534):1487–1491
Asai H, Ikezu S, Woodbury ME, Yonemoto GM, Cui L, Ikezu T (2014) Accelerated neurodegeneration and neuroinflammation in transgenic mice expressing P301L tau mutant and tau-tubulin kinase 1. Am J Pathol 184(3):808–818
Ikezu S, Ikezu T (2014) Tau-tubulin kinase. Front Mol Neurosci 7:33
Boillee S, Vande Velde C, Cleveland DW (2006) ALS: a disease of motor neurons and their nonneuronal neighbors. Neuron 52(1):39–59
Appel SH, Beers DR, Henkel JS (2010) T cell-microglial dialogue in Parkinson’s disease and amyotrophic lateral sclerosis: are we listening? Trends Immunol 31(1):7–17
Swarup V, Phaneuf D, Dupre N, Petri S, Strong M, Kriz J et al (2011) Deregulation of TDP-43 in amyotrophic lateral sclerosis triggers nuclear factor kappaB-mediated pathogenic pathways. J Exp Med 208(12):2429–2447
Huang C, Tong J, Bi F, Zhou H, Xia XG (2012) Mutant TDP-43 in motor neurons promotes the onset and progression of ALS in rats. J Clin Invest 122(1):107–118
Henkel JS, Beers DR, Zhao WH, Appel SH (2009) Microglia in ALS: the good, the bad, and the resting. J Neuroimmune Pharm 4(4):389–398
Philips T, Robberecht W (2011) Neuroinflammation in amyotrophic lateral sclerosis: role of glial activation in motor neuron disease. Lancet Neurol 10(3):253–263
Xiao Q, Zhao W, Beers DR, Yen AA, Xie W, Henkel JS et al (2007) Mutant SOD1(G93A) microglia are more neurotoxic relative to wild-type microglia. J Neurochem 102(6):2008–2019
Boillee S, Yamanaka K, Lobsiger CS, Copeland NG, Jenkins NA, Kassiotis G et al (2006) Onset and progression in inherited ALS determined by motor neurons and microglia. Science 312(5778):1389–1392
Beers DR, Henkel JS, Xiao Q, Zhao W, Wang J, Yen AA et al (2006) Wild-type microglia extend survival in PU.1 knockout mice with familial amyotrophic lateral sclerosis. Proc Natl Acad Sci U S A 103(43):16021–16026
Beers DR, Henkel JS, Zhao W, Wang J, Appel SH (2008) CD4+ T cells support glial neuroprotection, slow disease progression, and modify glial morphology in an animal model of inherited ALS. Proc Natl Acad Sci U S A 105(40):15558–15563
Hensley K, Fedynyshyn J, Ferrell S, Floyd RA, Gordon B, Grammas P et al (2003) Message and protein-level elevation of tumor necrosis factor alpha (TNF alpha) and TNF alpha-modulating cytokines in spinal cords of the G93A-SOD1 mouse model for amyotrophic lateral sclerosis. Neurobiol Dis 14(1):74–80
Liao B, Zhao W, Beers DR, Henkel JS, Appel SH (2012) Transformation from a neuroprotective to a neurotoxic microglial phenotype in a mouse model of ALS. Exp Neurol 237(1):147–152
Kawamura MF, Yamasaki R, Kawamura N, Tateishi T, Nagara Y, Matsushita T et al (2012) Impaired recruitment of neuroprotective microglia and T cells during acute neuronal injury coincides with increased neuronal vulnerability in an amyotrophic lateral sclerosis model. Exp Neurol 234(2):437–445
Marden JJ, Harraz MM, Williams AJ, Nelson K, Luo M, Paulson H et al (2007) Redox modifier genes in amyotrophic lateral sclerosis in mice. J Clin Invest 117(10):2913–2919
Frakes AE, Ferraiuolo L, Haidet-Phillips AM, Schmelzer L, Braun L, Miranda CJ et al (2014) Microglia induce motor neuron death via the classical NF-kappaB pathway in amyotrophic lateral sclerosis. Neuron 81(5):1009–1023
Kobayashi K, Imagama S, Ohgomori T, Hirano K, Uchimura K, Sakamoto K et al (2013) Minocycline selectively inhibits M1 polarization of microglia. Cell Death Dis 4:e525
Lewis KE, Rasmussen AL, Bennett W, King A, West AK, Chung RS et al (2014) Microglia and motor neurons during disease progression in the SOD1G93A mouse model of amyotrophic lateral sclerosis: changes in arginase1 and inducible nitric oxide synthase. J Neuroinflamm 11:55
Majerova P, Zilkova M, Kazmerova Z, Kovac A, Paholikova K, Kovacech B et al (2014) Microglia display modest phagocytic capacity for extracellular tau oligomers. J Neuroinflamm 11(1):161
Streit WJ, Braak H, Xue QS, Bechmann I (2009) Dystrophic (senescent) rather than activated microglial cells are associated with tau pathology and likely precede neurodegeneration in Alzheimer’s disease. Acta Neuropathol 118(4):475–485
Luo XG, Chen SD (2012) The changing phenotype of microglia from homeostasis to disease. Transl Neurodegener 1(1):9
Lucin KM, Wyss-Coray T (2009) Immune activation in brain aging and neurodegeneration: too much or too little? Neuron 64(1):110–122
Lee CK, Weindruch R, Prolla TA (2000) Gene-expression profile of the ageing brain in mice. Nat Genet 25(3):294–297
Sheng JG, Mrak RE, Griffin WS (1998) Enlarged and phagocytic, but not primed, interleukin-1 alpha-immunoreactive microglia increase with age in normal human brain. Acta Neuropathol 95(3):229–234
Ye SM, Johnson RW (1999) Increased interleukin-6 expression by microglia from brain of aged mice. J Neuroimmunol 93(1–2):139–148
Ye SM, Johnson RW (2001) An age-related decline in interleukin-10 may contribute to the increased expression of interleukin-6 in brain of aged mice. Neuroimmunomodulat 9(4):183–192
Sugama S, Yang L, Cho BP, DeGiorgio LA, Lorenzl S, Albers DS et al (2003) Age-related microglial activation in 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP)-induced dopaminergic neurodegeneration in C57BL/6 mice. Brain Res 964(2):288–294
Lee DC, Ruiz CR, Lebson L, Selenica MLB, Rizer J, Hunt JB et al (2013) Aging enhances classical activation but mitigates alternative activation in the central nervous system. Neurobiol Aging 34(6):1610–1620
Bachstetter AD, Norris CM, Sompol P, Wilcock DM, Goulding D, Neltner JH et al (2012) Early stage drug treatment that normalizes proinflammatory cytokine production attenuates synaptic dysfunction in a mouse model that exhibits age-dependent progression of Alzheimer’s disease-related pathology. J Neurosci 32(30):10201–10210
Hesse M, Modolell M, La Flamme AC, Schito M, Fuentes JM, Cheever AW et al (2001) Differential regulation of nitric oxide synthase-2 and arginase-1 by type 1/type 2 cytokines in vivo: granulomatous pathology is shaped by the pattern of L-arginine metabolism. J Immunol 167(11):6533–6544
Horowitz S, Binion DG, Nelson VM, Kanaa Y, Javadi P, Lazarova Z et al (2007) Increased arginase activity and endothelial dysfunction in human inflammatory bowel disease. Am J Physiol Gastrointest Liver Physiol 292(5):G1323–G1336
Ding H, Demple B (2000) Direct nitric oxide signal transduction via nitrosylation of iron-sulfur centers in the SoxR transcription activator. Proc Natl Acad Sci U S A 97(10):5146–5150
Acknowledgments
This work was supported by grants from the National Natural Sciences Foundation of China (No. 81171201) and the National Basic Research Program of China (No. 2011CB510003).
Conflict of Interest
The authors declare no conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Tang, Y., Le, W. Differential Roles of M1 and M2 Microglia in Neurodegenerative Diseases. Mol Neurobiol 53, 1181–1194 (2016). https://doi.org/10.1007/s12035-014-9070-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-014-9070-5