Abstract
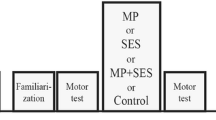
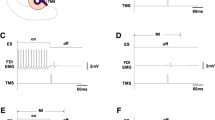
Rehabilitation with augmented electrical stimulation can enhance functional recovery after stroke, and cortical plasticity may play a role in this process. The purpose of this study was to compare the effects of three training paradigms on cortical excitability in healthy subjects. Cortical excitability was evaluated by analysing the input–output relationship between transcranial magnetic stimulation intensity and motor evoked potentials (MEPs) from the flexor muscles of the fingers. The study was performed with 25 healthy volunteers who underwent 20-min simulated therapy sessions of: (1) functional electrical stimulation (FES) of the finger flexors and extensors, (2) voluntary movement (VOL) with sensory stimulation, and (3) therapeutic FES (TFES) where the electrical stimulation augmented voluntary activation. TFES training produced a significant increase in MEP magnitude throughout the stimulation range, suggesting an increase in cortical excitability. In contrast, neither the FES nor voluntary movement alone had such an effect. These results suggest that the combination of voluntary effort and FES has greater potential to induce plasticity in the motor cortex and that TFES might be a more effective approach in rehabilitation after stroke than FES or repetitive voluntary training alone.





Similar content being viewed by others
References
Andersen B, Westlund B, Krarup C (2003) Failure of activation of spinal motoneurons after muscle fatigue in healthy subjects studied by transcranial magnetic stimulation. J Physiol 551:345–356
Cantello R, Gianelli M, Civardi C, Mutani R (1992) Magnetic brain stimulation: the silent period after the motor evoked potential. Neurology 42:1951–1959
Castro-Alamancos M, Garcia-Segura L, Borrell J (1992) Transfer of function to a specific area of the cortex after induced recovery from brain damage. Eur J Neurosci 4:853–863
Chae J, Yu D (1999) Neuromuscular stimulation for motor relearning in hemiplegia. Crit Rev Phys Med Rehabil Med 11:279–297
Chen R (2000) Studies of human motor physiology with transcranial magnetic stimulation. Muscle Nerve Suppl 9:26–32
Classen J, Liepert J, Wise SP, Hellett M, Cohen LG (1998) Rapid plasticity of human cortical movement representation induced by practice. J Neurophysiol 79:1117–1123
Devanne H, Lavoie BA, Capaday C (1997) Input-output properties and gain changes in the human corticopinal pathway. Exp Brain Res 114:329–338
Di Piero V, Chollet F, Mac Carthy P, Lenzi G, Frackowiak R (1992) Motor recovery after acute ischaemic stroke: a metabolic study. J Neurol Neurosurg Psychiatr 55:990–996
Ferster D (2004) Neuroscience. Blocking plasticity in the visual cortex. Science 303:1619–1621
Fuhr P, Cohen LG, Roth BJ, Hallet M (1991) Latency of motor evoked potentials to focal transcranial stimulation varies as a function of scalp position stimulated. Electroencephalogr Clin Neurophysiol 81(2):81–89
Gritsenko V, Prochazka A (2004) A functional electric stimulation-assisted exercise therapy system for hemiplegic hand function. Arch Phys Med Rehabil 85:881–885
Hermens HJ, Freriks B, Disselhorst-Klug C, Rau G (2000) Development of recommendations for SEMG sensors and sensor placement procedures. J Electromyogr Kinesiol 10(5):361–374
Ho KH, Nithi K, Mills KR (1998) Covariation between human intrinsic hand muscles of the silent periods and compound muscle action potentials evoked by magnetic brain stimulation: evidence for common inhibitory connections. Exp Brain Res 122:433–440
Inghilleri M, Berardelli A, Cruccu G, Manfredi M (1993) Silent period evoked by transcranial stimulation of the human cortex and cervicomedullary junction. J Physiol 466:521–534
Jacobs KM, Donoghue JP (1991) Reshaping the cortical motor map by unmasking latent intracortical connections. Science 251(4996):944–947
Jenkins WM, Merzenich MM (1987) Reorganisation of neocortical representations after brain injury: a neurophysiological model of the bases of recovery from stroke. In: Seil FJ, Herbert E, Carlson B (eds) Progress in brain research, vol 71. Elsevier, Amsterdam, p 249
Khaslavskaia S, Sinkjaer T (2005) Motor cortex excitability following repetitive electrical stimulation of the common peroneal nerve depends on the voluntary drive. Exp Brain Res 162:497–502
Khaslavskaia S, Ladouceur M, Sinkjaer T (2002) Increase in tibialis anterior motor cortex excitability following repetitive electrical stimulation of the common peroneal nerve. Exp Brain Res 145(3):309–315
Kido Thompson A, Stein RB (2004) Short-term effects of functional electrical stimulation on motor-evoked potentials in ankle flexor and extensor muscles. Exp Brain Res 159:491–500
Knash ME, Kido A, Gorassini M, Chan KM, Stein RB (2003) Electrical stimulation of the human common peroneal nerve elicits lasting facilitation of cortical motor-evoked potentials. Exp Brain Res 153:366–377
McDonnell MN, Ridding MC (2006) Afferent stimulation facilitates performance on a novel motor task. Exp Brain Res 170:109–115
Muellbacher W, Ziemann U, Boroojerdi B, Cohen L, Hallett M (2001) Role of the human motor cortex in rapid motor learning. Exp Brain Res 136:431–438
Nudo RJ (2003) Functional and structural plasticity in motor cortex: implication for stroke recovery. Phys Med Rehabil Clin N Am 14(1 Suppl):857–876
Nudo RJ (2007) Postinfarct cortical plasticity and behavioral recovery. Stroke 38(2 Suppl):840–845
Oliviero A, Profice P, Tonali PA, Pilato F, Saturno E, Dileone M, Ranieri F, Di Lazzaro V (2006) Effects of ageing on motor cortex excitability. Neurosci Res 55:74–77
Peckham PH, Knutson JS (2005) Functional electrical stimulation for neuromuscular applications. Annu Rev Biomed Eng 7:327–360
Perez MA, Lungholt BKS, Nyborg K, Nielsen JB (2004) Motor skill training induces changes in the excitability of the leg cortical area in healthy humans. Exp Brain Res 159:197–205
Pitcher JB, Miles TS (2002) Alterations in corticospinal excitability with imposed vs. voluntary fatigue in human hand muscles. J Appl Physiol 92:2132–2138
Popovic MB, Popovic DB, Sinkjaer T, Stefanovic A, Schwirtlich L (2002) Restitution of reaching and grasping promoted by functional electrical therapy. Artif Organs 26(3):271–275
Popovic MB, Popovic DB, Sinkjaer T, Stefanovic A, Schwirtlich L (2003) Clinical evaluation of functional electrical therapy in acute hemiplegic subjects. J Rehabil Res Dev 40:443–453
Popovic DB, Popovic MB, Sinkjaer T, Stefanovic A, Schwirtlich L (2004) Therapy of paretic arm in hemiplegic subjects augmented with a neural prosthesis: a cross-over study. Can J Physiol Pharmacol 82:749–756
Popovic MR, Thrasher TA, Adams ME, Takes V, Zivanovic V, Tonack MI (2006) Functional electrical therapy: retraining grasping in spinal cord injury. Spinal Cord 44(3):143–151
Powell J, Pandyan D, Granat M, Cameron M, Stott DJ (1990) Electrical stimulation of wrist extensors in poststroke hemiplegia. Stroke 30:1384–1389
Press WH, Flannery BP, Teukolsky SA, Vetterling WT (1986) Numerical recipes. Cambridge University Press, Cambridge
Ridding MC, Rothwell JC (1995) Reorganisation in human motor cortex. Can J Physiol Pharmacol 73(2):218–222
Ridding MC, Rothwell JC (1997) Stimulus/response curves as a method of measuring motor cortical excitability in man. Electroencephalogr Clin Neurophysiol 105:340–344
Ridding MC, Rothwell JC (1999) Afferent input and cortical organisation: a study with magnetic stimulation. Exp Brain Res 126:536–544
Ridding MC, Brouwer B, Miles TS, Pitcher JB, Thompson PD (2000) Changes in muscle responses to stimulation of the motor cortex induced by peripheral nerve stimulation in human subjects. Exp Brain Res 131:135–143
Rossini PM, Pauri F (2000) Neuromagnetic integrated methods tracking human brain mechanisms of sensorimotor areas ‘plastic’ reorganisation. Brain Res Rev 33:131–154
Rosenkranz K, Rothwell JC (2004) The effect of sensory input and attention on the sensorimotor organization of the hand area of the human motor cortex. J Physiol 561(Pt 1):307–320
Sacco P, Newberry R, McFadden L, Brown T, McComas AJ (1998) Depression of human electromyographic activity by fatigue of synergistic muscle. Muscle Nerve 20:710–717
Seitz RJ, Bütefisch CM, Kleiser R, Hömberg V (2004) Reorganisation of cerebral circuits in human ischemic brain disease. Restor Neurol Neurosci 22(3–5):207–229
Siebner HR, Rothwell JC (2003) Transcranial magnetic stimulation: new insights into representational cortical plasticity. Exp Brain Res 148:1–16
Smith CD, Umberger GH, Manning EL, Slevin JT, Wekstein DR, Schmitt FA, Markesbery WR, Zhang Z, Gerhardt GA, Kryscio RJ, Gash DM (1999) Critical decline in fine motor hand movements in human ageing. Neurology 53(7):1458–1461
Stetkarova I, Leis AA, Stokic DS, Delapasse JS, Tarkka IM (1994) Characteristics of the silent period after transcranial magnetic stimulation. Am J Phys Med Rehabil 73:98–102
Taylor JL, Allen GM, Butler JE, Gandevia SC (1997) Effect of contraction strength on responses in biceps brachii and adductor pollicis to transcranial magnetic stimulation. Exp Brain Res 117:472–478
Taylor JL, Butler JE, Gandevia SC (1999) Altered responses of human elbow flexors to peripheral-nerve and cortical stimulation during a sustained maximal voluntary contraction. Exp Brain Res 127:108–115
Thrasher TA, Flett HM, Popovic MR (2006) Gait training regimen for incomplete spinal cord injury using functional electrical stimulation. Spinal Cord 44(6):351–367
Winslow J, Jacobs PL, Tepavac D (2003) Fatigue compensation during FES using surface EMG. J Electromyogr Kinesiol 13:555–568
Acknowledgements
The authors wish to thank the participants of the study and the personnel of the Centre of Sensory-Motor Interaction, Aalborg University, for their help during the study. This study was supported by grants from the Danish National Research Foundation and Det Obelske Familiefond.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Barsi, G.I., Popovic, D.B., Tarkka, I.M. et al. Cortical excitability changes following grasping exercise augmented with electrical stimulation. Exp Brain Res 191, 57–66 (2008). https://doi.org/10.1007/s00221-008-1495-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00221-008-1495-5