Article Text
Abstract
Background In patients with a minor ischaemic stroke or transient ischaemic attack (TIA), separate trials have shown that dual antiplatelet therapy with clopidogrel plus aspirin (clopidogrel–aspirin) or ticagrelor plus aspirin (ticagrelor–aspirin) are more effective than aspirin alone in stroke secondary prevention. However, these two sets of combination have not been directly compared. Since clopidogrel was less effective in stroke patients who were CYP2C19 loss-of-function (LOF) allele carriers, whether ticagrelor–aspirin is clinically superior to clopidogrel–aspirin in this subgroup of patients with stroke is unclear.
Aim To describe the rationale and design considerations of the Clopidogrel in High-risk patients with Acute Non-disabling Cerebrovascular Events (CHANCE-2) trial.
Design CHANCE-2 is a randomised, double-blind, double-dummy, placebo-controlled, multicentre trial that compares two dual antiplatelet strategies for minor stroke or TIA patients who are CYP2C19 LOF allele carriers: ticagrelor (180 mg loading dose on day 1 followed by 90 mg twice daily on days 2–90) or clopidogrel (300 mg loading dose on day 1 followed by 75 mg daily on days 2–90), plus open-label aspirin with a dose of 75–300 mg on day 1 followed by 75 mg daily on day 2–21. All will be followed for 1 year.
Study outcomes The primary efficacy outcome is any stroke (ischaemic or haemorrhagic) within 3 months and the primary safety outcome is any severe or moderate bleeding event within 3 months.
Discussion The CHANCE-2 trial will evaluate whether ticagrelor–aspirin is superior to clopidogrel–aspirin for minor stroke or TIA patients who are CYP2C19 LOF allele carriers.
Trial registration number NCT04078737.
- stroke
Data availability statement
Data sharing not applicable as no datasets generated and/or analysed for this article.
This is an open access article distributed in accordance with the Creative Commons Attribution Non Commercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited, appropriate credit is given, any changes made indicated, and the use is non-commercial. See: http://creativecommons.org/licenses/by-nc/4.0/.
Statistics from Altmetric.com
Introduction and rationale
In patients with acute non-disabling cerebrovascular events, including those with acute minor stroke or transient ischaemic attack (TIA), the subsequent recurrent stroke and cardiovascular events are more than 6% in the first year.1 2 As shown in Clopidogrel in High-risk patients with Acute Non-disabling Cerebrovascular Events (CHANCE) trial and the Platelet-Oriented Inhibition in New TIA and Minor Ischaemic Stroke (POINT) trial, a lower risk of stroke recurrence was observed with a combination of clopidogrel and aspirin (clopidogrel–aspirin) than with aspirin alone.3 4 However, the choice of an optimal dual antiplatelet therapy regimen for patients with strokes requires a tailored approach based on the characteristics of the patient. The genetic substudy of the CHANCE trial showed that clopidogrel plus aspirin reduced the risk of stroke recurrence in non-carriers of the cytochrome P450 2C19 (CYP2C19) loss-of-function (LOF) alleles compared with aspirin alone but not in carriers. Therefore, clopidogrel may not provide additional benefit in stroke prevention compared with aspirin alone for the carriers.5 More recently, the substudy of POINT failed to reproduce the above findings as seen in the CHANCE trial, possibly from a limited statistical power.6 In addition, a meta-analysis of 15 studies that included 4762 patients with stroke or TIA found that carriers of CYP2C19 LOF alleles were at higher risk of vascular events than non-carriers when taking clopidogrel.7
Ticagrelor is a reversible oral antagonist directly blocking the P2Y12-adenosine diphosphate receptor that needs no catabolite activation, which in turn yields greater mean levels of platelet inhibition than clopidogrel.8 9 From the Acute Stroke or Transient Ischaemic Attack Treated with Aspirin or Ticagrelor and Patient Outcomes trial involving participants with acute ischaemic stroke or TIA, ticagrelor was not found to be more effective than aspirin in reducing the risk of 90-day vascular events.5 However, among the Asian patients, a tendency towards better efficacy was seen in a subgroup analysis.10 The Platelet Reactivity in Acute Stroke or Transient Ischaemic Attack trial showed that participants with ischaemic stroke or TIA who were treated with ticagrelor plus aspirin (ticagrelor–aspirin) had a lower proportion of high platelet reactivity than those who were treated with clopidogrel–aspirin, particularly in the those who were CYP2C19 LOF allele carriers.11 The Acute Stroke or Transient Ischaemic Attack Treated with Ticagrelor and Aspirin for Prevention of Stroke and Death trial showed that dual antiplatelet therapy (ticagrelor–aspirin) was better than mono antiplatelet therapy (aspirin) for secondary stroke prevention in people with mild-to-moderate acute ischaemic stroke or TIA, but the rate of haemorrhage was also elevated.12
While separate trials have shown similar treatment effects, no direct comparison has been conducted in this patient population. In some populations, more than 50% of them carry CYP2C19 LOF alleles so that the conversion to an active form of clopidogrel is reduced. We hence hypothesised that, compared with clopidogrel–aspirin, ticagrelor–aspirin may favourably impact the clinical outcomes in high-risk participants with acute non-disabling cerebrovascular events and were carriers of the CYP2C19 LOF allele. Therefore, the CHANCE-2 trial was designed to test such hypothesis. The intention of this trial is to evaluate the safety and efficacy of ticagrelor–aspirin versus clopidogrel–aspirin for minor stroke or high-risk TIA patients who were CYP2C19 LOF allele carriers. This article describes the design of the CHANCE-2 trial and is a summary of protocol.
Methods
Design and patients population
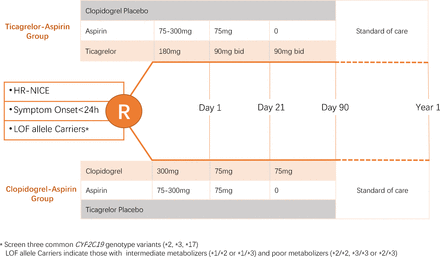
CHANCE-2 is a randomised, double-blind, double-dummy, placebo-controlled multicentre trial. Patients with CYP2C19 LOF alleles are randomised with 1:1 to one of the two strategies (ticagrelor–aspirin or clopidogrel–aspirin) within 24 hours of the onset of cerebrovascular Events. All participants will be followed for 3 months on study intervention, with another 9 months of follow-up on standard of care. The study design of the CHANCE-2 is shown in figure 1. The trial will enrol subjects age ≥40 years with acute non-disabling ischaemic stroke, with a National Institutes of Health Stroke Scale (NIHSS) score ≤3 or high-risk TIAs (ABCD2 score ≥4). Once randomised, they will be treated with study drug within 24 hours of symptoms onset. Figure 2 lists the summary of inclusion and exclusion criteria. Prior to initiating the study, each clinical site obtains institutional review board (IRB) approval for the protocol, informed consent, and materials used to recruit subjects. Participants from 240 hospitals in China will be enrolled in CHANCE-2.
CHANCE-2 study design. CHANCE-2, Clopidogrel in High-risk patients with Acute Non-disabling Cerebrovascular Events; NIHSS, National Institutes of Health Stroke Scale; TIA, transient ischaemic attack;ASA,aspirin; ULN, upper limit of normal; ALT, alanine aminotransferase; AST,aspartate transaminase;AV, atrioventricular.
Summary of inclusion and exclusion criteria. HR-NICE, high-risk patients with acute non-disabling cerebrovascular events (TIA or acute minor stroke); LOF, loss-of-function.
Genotyping
In this study, a novel point-of-care genetic test platform will be used to identify carriers of CYP2C19 LOF alleles including poor metabolisers with at least two *2 or *3 alleles (*2/*2, *2/*3, or *3/*3) or intermediate metabolisers with one *2 or *3 allele (*1/*2 or *1/*3). The CYP2C19 genotyping will be implemented by the GMEX Point-of-Care Genotyping system, including a portable DNA analyser, genotyping reagents, and a buccal sample collection kit. The system uses non-invasive sampling by buccal swab and integrates automated steps of DNA extraction, PCR-based amplification, fluorescent signal detection and genotype determination. The system integrates controls to monitor the performance of a run and ensure ongoing quality from the GMEX system. The analysis includes the single nucleotide polymorphisms CYP2C19*2 (681G>A, rs4244285), CYP2C19*3 (636G>A, rs4986893), and CYP2C19*17 (−806C>T, rs12248560), which will be genotyped on screening. The average result turn-around-time for the method of GMEX is 85 min.13
Randomisation
Participants will be randomised (1:1) to receive either ticagrelor–aspirin or clopidogrel–aspirin. A randomisation sequence will be generated centrally using random-permuted fixed-size blocks methods from the Statistics and Data Centre at the China National Clinical Research Centre for Neurological Diseases. The randomisation computer program makes the treatment assignment based on the current status of treatment group distribution within each clinical centre as well as overall balance of treatment assignment. Following randomised allocation, the study intervention will be administered to the patient as early as possible.
Intervention
Eligible participants are randomly assigned to one of two arms:
Ticagrelor–aspirin group
A 180 mg loading dose of Ticagrelor on day 1, followed by 90 mg two times per day on days 2–90 plus a 75–300 mg loading dose of aspirin, followed by 75 mg daily for 21 days;
Clopidogrel–aspirin group
A 300 mg loading dose of clopidogrel on day 1, followed by 75 two times per day on days 2–90 plus a 75–300 mg loading dose of aspirin followed by 75 mg daily for 21 days.
Patient will take the first dose of the study drug on the first visit. Then, for the next 12 weeks, the study drug should be taken twice daily, 12 hours apart. Once the study period is over at the end of 12th week, the treating physician will decide what antiplatelet drug to be continued. All participants will be followed for 1 year.
Primary outcomes
The primary efficacy outcome is any stroke (ischaemic or haemorrhagic) within 3 months. Definitions of stroke are provided in online supplemental table 1.
Supplemental material
Secondary outcomes
Secondary outcomes include the following events: (1) Any new stroke events within 30 days and 1 year; (2) New clinical vascular events including stroke, TIA, myocardial infarction and vascular deaths within 3 months and 1 year ; (3) New ischaemic stroke within 3 months and 1 year; (4) Disabling stroke (Modified Rankin Scale score, mRS >1) at 3 months and 1 year; (5) Incidence and severity of recurrent stroke and TIA during follow-up to 3 months and 1 year. Severity is measured using a six-level ordered categorical scale that incorporates the mRS: fatal stroke/severe non-fatal stroke (mRS 4 or 5)/moderate stroke (mRS 2 or 3)/mild stroke (mRS 0 or 1)/TIA/no stroke-TIA; (6) Neurological impairment at 3 months (NIHSS increased ≥4 from baseline); (7) Quality of Life (EuroQol-5dimension) scale. The influence on treatment effect of age, gender, body mass index, index event type (TIA vs minor stroke), time from index event to randomisation, aetiology subtype, diabetes mellitus, hypertension, type of LOF allele, previous ischaemic stroke or TIA, prior antiplatelet therapy, prior statin therapy, prior smoking status and symptomatic intracranial and extracranial artery stenosis will be evaluated in subgroup analyses.
Safety outcomes
The primary safety outcome is severe or moderate bleeding event within 3 months. The bleeding event is defined by the criteria from the Global Utilisation of Streptokinase and Tissue Plasminogen Activator for Occluded Coronary Arteries (GUSTO) trial.14 Severe haemorrhage is defined as fatal or intracranial haemorrhage or other haemorrhage causing haemodynamic compromise that requires blood or fluid replacement, inotropic support or surgical intervention. Moderate haemorrhage is defined as bleeding that required transfusion of blood but does not lead to haemodynamic compromise requiring intervention. Other secondary safety outcomes include the following events within a 3-month and 1-year time frame: (1) Incidence of severe bleedings or moderate bleedings (GUSTO definition) at 1 year; (2) Bleeding events; (3) Mortality; (4) Adverse events (AEs)/serious adverse events (SAEs) reported by the investigators.
Data safety and monitoring board
To ensure ethically conduct of the trial and patient safety, the data safety and monitoring board (DSMB) will meet per protocol and monitor the progress of the trial. Members of DSMB do not participate in the trial and their responsibilities are defined by the trial Executive Committee prior to the beginning of the trial. DSMB will provide their recommendations in written statement to the Chairs of trial Steering Committee after each meeting.
Sample size
The sample size calculation is based on the rate of the primary outcome (3-month risk of stroke) and estimated effect size. We used the following assumptions: (1) Significance level of 0.048 for a two-sided test; (2) Statistical power of 90%; (3) 3-month rate of stroke event is 9.4% in control group based on the data from the CHANCE trial5; (4) Proportional risk reduction of 25% (Rate ratio=0.75); (5) of 5% over 3 months. We estimated that 6396 eligible patients (3198 for each treatment group) are required. As 58.8% of patients are CYP2C19 LOF allele carriers,5 we need to screen about 10 878 patients in total.
Interim analysis
The interim analysis will focus on patient recruitment, baseline comparability of treatment arms, sample sizes with regard to event rates, loss to follow-up, adverse effects data and effect of treatment on the primary endpoints. We plan one interim analysis when 60% of total patients have undergone randomisation and completed the follow-up. The sample size is inflated to account for one interim analysis of the primary efficacy outcome with the use of an O’Brien-Fleming spending function, and a p<0.008 is considered to be statistically significant during the interim analysis. In the final analysis, a p<0.048 is considered to be statistically significant in the current study. Trial investigators will be blinded to the interim outcome results.
Statistical analyses
An intention-to-treat analysis will be used for all participants randomised to an intervention group. Participants will be censored at their last follow-up assessment when experiencing a clinical event, at the end of study, or at the time of withdrawal from the study. Statistically, the cumulative risk of any ischaemic or haemorrhagic event will be reported as a Kaplan-Meier estimates during the 90-day follow-up. Cox proportional hazards methods will be used for HR calculation at 95% CIs. The treatment effect will be assessed by the log-rank test. This approach is to maximise the time-dependent information in the trial while still acknowledge the ease of interpretation of risks. Accounting for a single interim analysis, a p value of 0.048 will be considered to indicate statistical significance for the primary outcome. Detailed analysis plans will be given in the statistical analysis plan before the database is locked and the blind is broken.
Study organisation
The Trial Steering Committee provides oversight and strategic input and will meet twice yearly. The Trial Management Committee runs the trial on a day-to-day basis and is based at the CHANCE-2 Trial Coordinating Centre of the China National Clinical Research Centre for Neurological Diseases. Outcomes, SAEs, and brain imaging are adjudicated by trained assessors masked to treatment assignment.
Discussion
CHANCE-2 will address a major issue in secondary prevention in those TIA or minor stroke patients who are CYP2C19 LOF allele carriers. Furthermore, the safety and efficacy of ticagrelor–aspirin or clopidogrel–aspirin will be assessed.
Two recent RCTs revealed that the short term clopidogrel–aspirin therapy is beneficial after acute minor stroke or TIA.3 4 However, several studies have showed the higher risk of poor clinical outcomes in CYP2C19 LOF allele carriers who receiving clopidogrel.15–18 A genetic substudy of the Secondary Prevention of Small Subcortical Strokes trial reported that patients with one CYP2C19 LoF allele carriers had higher odds of stroke recurrence compared with non-carriers.19 A preplanned substudy of the CHANCE trial found that CYP2C19 LoF carriers had 1.5 times the risk of recurrent stroke as non-carriers at 90 days.5 To date, there is no evidence from randomised trials for a direct comparison of treatment effect between ticagrelor and clopidogrel with aspirin added on for secondary stroke prevention. Clopidogrel–aspirin is still the approved option for secondary stroke prevention in patients with acute non-disabling cerebrovascular events.20
Summary and conclusions
The CHANCE-2 trial will produce reliable data on whether ticagrelor–aspirin is superior to the recommendations from current guidelines, clopidogrel–aspirin, for the prevention of recurrent stroke in patients who already have experienced an acute minor stroke or high-risk TIA and are carriers of CYP2C19 LOF alleles.
Data availability statement
Data sharing not applicable as no datasets generated and/or analysed for this article.
Ethics statements
Ethics approval
The CHANCE-2 trial was approved by ethics committee at Beijing Tiantan Hospital (IRB approval number: KY2019-035-02) and all participating centres
References
Supplementary material
Supplementary Data
This web only file has been produced by the BMJ Publishing Group from an electronic file supplied by the author(s) and has not been edited for content.
Footnotes
Twitter @yilong
Contributors YW, CJ, PMB, A-DX and QD contributed to the design of the study and contribute to its oversight. HL, XM, XX, JJ, AW and YP coordinated the study. YW and XX wrote the first draft of the manuscript, which was edited by all other authors.
Funding The Ministry of Science and Technology of the People’s Republic of China (MOST), Beijing Municipal Science and Technology Commission and Chinese Stroke Association (CSA) fund CHANCE-2. This work was supported by grants from National Science and Technology Major Project (2017ZX09304018) and Beijing Municipal Science and Technology Commission (D171100003017002). Salubris contributes ticagrelor, clopidogrel and its placebo at no cost and with no restrictions. The principal investigator and executive committee will have full access to the entire dataset at trial completion and are responsible for analysis and publication in collaboration with the sponsor.
Competing interests None declared.
Provenance and peer review Not commissioned; externally peer reviewed.