Article Text
Statistics from Altmetric.com
Strengths and limitations of this study
This is the first prospective registry study to predict the prognosis of intracranial dissecting aneurysm with intramural haematoma (IMH) according to evaluation of neovascularisation by in vivo imaging. The advanced dynamic contrast-enhanced MRI sequence will be used to evaluate the neovascularisation in IMH. The main limitation of this study is that the findings of this study would be lack of validation with histological findings.
Background
Intracranial dissecting aneurysm (IDA) is a challenging disease that can result in stroke in young and middle-aged adults.1 2 Intramural haematoma (IMH), one of the typical imaging findings of IDA, can grow continuously in untreated IDA.3 4 The IMH is usually located within the layers of the tunica media and formed by the blood under arterial pressure entering the wall of the artery from the intimal tear.5 Due to the relatively high complication rate associated with surgical procedures, endovascular treatments have become the first-line therapy for such lesions.6 According to whether the parent artery is maintained, endovascular treatment is divided into deconstructive (proximal arterial occlusion and internal trapping) or reconstructive (stent implantation with or without coiling) techniques.7 However, recanalisation of IDA is a great challenge for endovascular treatment, and influences the prognosis of patients.8
The formation of an IMH is a critical event in the progression of IDA, and IDA may grow because of recurrent IMH, even after deconstructive endovascular treatment4 9; such increased IMH size can result in severe compressive symptoms or even death.10 At present, the mechanism of the continuous growth of IMH after endovascular treatment of IDA is still unclear. Some authors believe that continuous haemorrhage of the vasa vasorum in the IMH results in continuous enlargement of the IMH.9 11 However, this theory has not been proved by in vivo imaging.
Dynamic contrast-enhanced MRI (DCE-MRI) has been widely used as a powerful tool to detect and assess the spatial and temporal heterogeneity of tumour neovascularisation.12 13 Similarly, the vasa vasorum in the IMH can also be evaluated by DCE-MRI. In this observational study, we hypothesise that the vasa vasorum plays an important role in the recanalisation of IDA with IMH after endovascular treatment. Therefore, examining the vasa vasorum in the IMH by DCE-MRI may facilitate accurate prediction of the prognosis of IDA with IMH, and would potentially be of great value in clinical practice. In the future, DCE-MRI analysis could provide a theoretical basis for the prognosis and optimal treatment strategy of the disease.
Methods and design
DCE-MRI analysis for prognosis of IDA with IMH after endovascular treatment (DEMAT) is a prospective observational registry study. It is sponsored by the National Nature Science Foundation of China, and will be performed at Beijing Tiantan Hospital. Patients with IMH in IDA treated by endovascular treatment will be recruited. The treatment strategy will not be influenced by the patients’ participation in this observational study. Participating patients or their relatives will be informed that the anonymous collection of their data and the privacy of their data will be protected. They will be able to withdraw from the study at any time. The protocol conforms to the Standard Protocol Items: Recommendations forInterventional Trials (SPIRIT) 2013 statement, and the SPIRIT checklist.14 A flow chart of the study protocol is shown in figure 1.
Flow chart of the protocol. DCE-MRI, dynamic contrast-enhanced MRI; IDA, intracranial dissecting aneurysm; IMH, intramural haematoma.
Aims
DEMAT is a single-centre prospective study. It is designed to evaluate the risk factors of recanalisation of IDA with IMH, especially to determine the association between the density of the vasa vasorum in IMH and the outcome of IMH changes, as observed on DCE-MRI. In the future, the results of this study maybe could provide a theoretical basis for the prognosis and intervention of such lesions, such as that the antiangiogenesis therapeutics may be an option for this lesion.
Design
This study will establish a prospective cohort of patients who had an IDA with IMH and received endovascular treatment. All the patients will undergo DCE-MRI before and 6 months after the endovascular treatment. According to the follow-up results of the DCE-MRI, the IDAs will be divided into two groups: an IMH stabilisation group and an IMH enlargement group. Then, quantitative DCE-MRI analysis of the vasa vasorum in the IMH will be performed, and differences between the two groups will be compared to evaluate the feasibility of early prediction of IMH in IDA by DCE-MRI.
Recruitment criteria
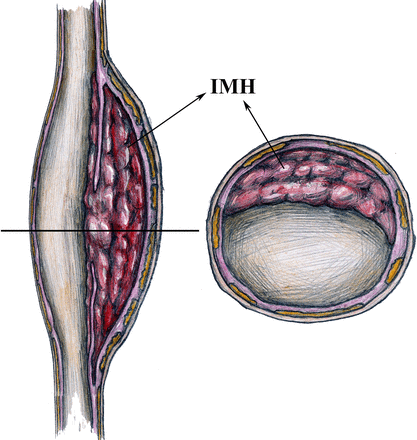
The inclusion criteria are: adult patients (age 18–80 years); an ability to cooperate during the MRI examination; patients who are diagnosed with IDA according to digital subtraction angiography and MRI; expectation of adequate patient safety during the examinations; unequivocal evidence of IDA with IMH on MRI (IMH>5 mm in the perpendicular plane to the long axis of the vessel)(figure 2); IDA treated by the endovascular approach; and the patients’ willingness to participate in the study.
Intracranial dissecting aneurysm with intramural haematoma (IMH) in the layers of the tunica media (illustrated by Yisen Zhang): perpendicular view to the long axis of the vessel (left) and perpendicular view to the short axis of the vessel (right).
The exclusion criteria are: patients with pre-existing diagnoses of arteritis, fibromuscular dysplasia, iatrogenic aneurysms or pseudoaneurysms; extracranial dissecting aneurysms extended into the intracranial segment; patients with other diseases or poor general condition with expected survival of less than 2 years; IDA that has already been treated by endovascular treatment; and patients lacking DCE-MRI follow-up.
Data collection
The following information will be collected and recorded. Patient baseline information included age, sex, clinical presentation, comorbidities (hypertension, diabetes, hyperlipidaemia), history of cigarette smoking and alcohol drinking, and familial history of aneurysms. IDA characteristics included size, shape, location, ruptured or unruptured, the number of aneurysms and their maximum size. Treatment-related data included treatment therapy (coiling alone, balloon or stent-assisted coiling, or flow diverter treatment), the number of devices and angiographic results. DCE-MRI related parameters including contrast extravasation rate (transfer constant (Ktrans)), the extracellular extravascular fractional volume (Ve), the fractional plasma volume (Vp), the region of interest (ROI) concentration of contrast agent (C(t)) and arterial input function (Cp(t)) were collected. Immediate angiographic results after the endovascular procedure and angiographic results at 6-month follow-up were collected. Perioperative complications will also be recorded, and throughout the duration of follow-up, the modified Rankin Scale (mRS) will be used to assess the patients’ degree of disability, with an mRS score of 0–2 being considered a favourable outcome, and a score of 3–6 an unfavourable outcome.
An independent angiographic committee consisting of two radiologists with more than 10 years of experience in vascular neuroimaging and one radiologist with more than 15 years of experience in vascular neuroimaging will be set up to evaluate the progress of the occlusion of aneurysms and measure the maximum size of the IMH. The independent angiographic committee will be blinded to the clinical data. First, the two radiologists with more than 10 years of experience in vascular neuroimaging will review all cases and determine the affected vessels, occlusion grade of aneurysm, and the maximum size of IMH on axial, coronal and sagittal MR images at the level of the lesion’s largest diameter on both pretreatment and follow-up MR images. If there are any discrepant findings between the two readers, a final diagnosis will be made by the radiologist with more than 15 years of experience in vascular neuroimaging. The occlusion grade of aneurysms will be classified as ‘complete occlusion’ if the aneurysmal sac is not filled with contrast media after the endovascular procedure, ‘residual neck’ if contrast agent is still present in the aneurysmal neck and ‘residual aneurysm’ if contrast agent is still present in the aneurysmal sac. According to the preoperative and follow-up DCE-MRI results, the IDAs will be divided into two groups: the IMH stabilisation group and the IMH enlargement group. IMH enlargement is defined as either growth ≥2.0 mm in at least one direction among the maximum axial, coronal and sagittal diameters, or growth ≥1.0 mm in two directions among the maximum axial, coronal and sagittal diameters.15
Imaging protocol
All patients will be scanned on 3 T MRI scanners (Achieva TX; Philips Healthcare, Best, the Netherlands) using 32-channel head coils. Scanning sequences will include standard DCE-MRI, 3D time-of flight (3D-TOF) imaging and precontrast and postcontrast 3D black-blood T1-weighted volume isotropic turbo spin-echo acquisition sequences (3D T1-VISTA). The 3D-TOF imaging will be performed first, to locate the target IDA. The 3D T1-VISTA will then be performed to acquire precontrast vessel wall images. The precontrast T1 mapping, DCE-MRI and postcontrast 3D T1-VISTA will then be acquired in turn.
The sequence parameters will be set following those used in previous studies.16 For the 3D-TOF, they will include: repetition time (TR)/echo time (TE)=25/3.5 ms; flip angle=20°; field of view (FOV)=200 × 200×84 mm; and reconstructed voxel size=0.35 × 0.35×0.7 mm. For the precontrast 3D T1-VISTA, they will include: TR/TE=700/30 ms; turbo spin echo factor=49; FOV=160 × 160×54 mm; voxel size=0.6 × 0.6×0.6 mm; and 90 transverse slices. Sparse-enforced sensitivity encoding parallel imaging technique with an acceleration factor equal to 2 will be applied to accelerate the imaging process. Precontrast T1-mapping will be performed using a 3D variable flip angle (VFA) sequence with optimised flip angles of 2°, 4°, 9° and 25°. To improve the signal-to-noise ratio and ensure the accuracy of the measurement, the signal will be collected three times. B1-mapping will be used to correct variations in the VFA. DCE-MRI will be acquired using a 3D T1-weighted spoiled gradient-echo sequence. The total DCE-MRI scan time will be about 6 min, and the time resolution 10 s. At the fifth frame of the dynamic scan, a dose of 0.1 mmol/kg gadolinium-diethylenetriamine pentaacetic acid (Gd-DTPA; Magnevist; Bayer HealthCare Pharmaceuticals, Wayne, New Jersey) will be intravenously injected at a rate of 1.5 mL/s, followed by a 20 mL saline flush delivered at the same rate. The DCE-MRI scan parameters will include: TR/TE=3.9/2 ms; flip angle=15°; FOV=160 × 160 mm; spatial resolution=1 × 1 mm; slice thickness=2 mm; number of slices=15. After the DCE-MRI sequence is completed, the 3D T1-VISTA will be repeated to obtain postcontrast vessel wall images. The DCE-MRI and 3D T1-VISTA will be performed at the same location.
Imaging analysis
The DCE-MRI analysis will be performed at the Department of Biomedical Engineering, Tsinghua University. The engineers and radiologists performing the analysis will be blinded to the clinical data. For the DCE-MRI analysis, the engineers performed the generation of Ktrans and Vp maps. Then, the radiologists place an ROI on the IMH and got the values of Ktrans and Vp within the ROI. A permeability-limited modified Tofts model will be used to quantify the pharmacokinetics of the contrast agent in the DCE-MR imaging, according to the following relationship16 17:
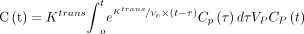
where Ktrans is the contrast agent transfer rate from the intravascular space to the extracellular extravascular space in units per minute (which reflects the permeability of neovascularisation); Ve is the extracellular extravascular fractional volume; Vp is the fractional plasma volume (which reflects the density of neovascularisation); C(t) is the contrast agent concentration of the ROI; and Cp(t) is the arterial input function.18 In this study, the contrast agent concentration will be calculated from the signal intensity values in the DCE-MRI and precontrast T1 mapping data, while the arterial input function will be taken from the concentration of contrast agent in the aneurysm. The modified Tofts model will be applied to each pixel in each slice of the imaging volume. The Ktrans and Vp maps will be generated by the least-squares method.
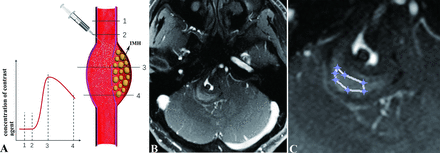
To evaluate IMH neovascularisation and permeability, two radiologists with more than 10 years of experience, who will be blinded to the clinical data of the patients, will review the DCE-MRI of all cases independently. Selecting the slice showing the highest Ktrans value for the IMH, they will place an ROI with a minimum of 10 voxels on the IMH (figure 3). A reference ROI will be placed on a normal intracranial artery on the same slice. IMH neovascularisation and permeability will be evaluated as Ktrans and Vp within the ROI, excluding voxels with blood signal contamination (Vp>0.5). The values of Ktrans and Vp from these two radiologists will be averaged. If there is significant disagreement between the two radiologists, the data will be evaluated by the third radiologist with more than 15 years of experience in vascular neuroimaging.
Dynamic contrast-enhanced MRI (DCE-MRI) analysis for the intracranial dissecting aneurysm with intramural haematoma (IMH). (A) If the IMH was rich of vasa vasorum, the IMH would gather more contrast agent after the contrast agent injected due to the permeability of vasa vasorum (illustrated by Yisen Zhang). (B) The slice of DCE-MRI images showing the highest value of DCE-MRI related parameters for the IMH was selected. (C) A region of interest was placed on the IMH of the selected slice to get the averaged values of DCE-MRI parameters.
Sample size and data analysis
According to our previous data, the ratio of the IMH stabilisation group to the IMH enlargement group should be approximately 3 to 1. The mean Ktrans of the IMH stabilisation group was 0.0262±0.0185/min, while in the IMH enlargement group it was 0.0516±0.0330/min. A sample size of 68 patients would give a power of 80% with a two-sided significance level of p=0.05. Considering that about 10% of patients are likely to be lost in follow-up, a sample size of 76 patients will need to be recruited, and a total of 80 patients will be enrolled.
The one-sample Kolmogorov-Smirnov test will be used to identify normally distributed variables. Continuous variables will be presented as mean and SD or median (IQR), as appropriate. Categorical variables will be expressed as count (percentage). The paired Student’s t-tests or Wilcoxon signed rank test will be used to compare Ktrans and Vp between IMH and reference normal artery. One-way analysis of variance or the Mann-Whitney U-test will be used to compare Ktrans and Vp among the different groups. Univariate analysis of other factors, including the characteristics of the patients, procedures and IDAs, will be performed to examine the associations of IMH growth with other potential risk factors, with Student’s t-test, the Mann-Whitney U-test, the χ2 test, or Fisher’s exact test being used as appropriate. All data processing and statistical analyses will be performed using SPSS V.22.0 (IBM Corp). Statistical significance is defined as p<0.05.
Data management
All data will be collected on written and electronic case report forms (CRF). The electronic CRFs will then be input into a study database. Yisen Zhang is the principal investigator and performed the specified regular monitoring of data safety and accuracy in a timely manner during the observational study. He will also supervise the management of all data interpretation, statistical analysis and results dissemination. Any serious adverse events will be recorded. The serious adverse events were referred to the undesirable experience related to the DCE-MRI examination and the use of contrast agent, which resulted in any change form baseline symptoms, such as allergic bronchospasm, seizures, permanent deficit, prolongation of hospitalisation, life threatening or even death. If any issues arise, the principal investigator and Institutional Review Board of Beijing Tiantan Hospital can decide to terminate the study.
Duration of the study
A total of 80 patients with IMH in IDA treated by endovascular therapy in Beijing Tiantan Hospital and meeting the inclusion and exclusion criteria will be enrolled in this observational study. Study recruitment started in June 2019, and the estimated primary completion date is July 2021.
Discussion
Recanalisation has posed a great challenge to endovascular treatment of IDAs.19–21 For IDA with IMH, the IMH plays an important role in the recanalisation.4 In a previous study, IDA with IMH sometimes continued to grow, even after the parent arteries had been sacrificed.10 Moreover, the underlying mechanisms are still unclear. Some authors hypothesise that the recurrent IDA with IMH results from rupture of the vasa vasorum.9 11 Neovascularisation in the IMH seems to be fragile, and could cause repetitive haemorrhage in the IMH, which could result in chronic enlargement of the IMH. However, this theory has not been validated by in vivo imaging.
In some previous studies, DCE-MRI was used to quantify the microvasculature of tumour and arterial wall according to the transfer constant Ktrans12 22 23, and Ktrans values were found to be associated with neovasculature on subsequent histopathological analyses.22 24 Nakatomi et al found neovascularisation in the IMH of IDA in a postmortem study, which indicates the feasibility of using DCE-MRI to quantify the vasa vasorum in IMH.4 Neovascularisation in the IMH could be used as a potential marker of IMH instability, as the enlargement of IMH may be a result of repetitive haemorrhage of the vasa vasorum. If this is the case, we could use DCE-MRI to predict the outcome of IMH after endovascular treatment, thus predicting the prognosis for IDA. Our study will be the first prospective observational registry study to predict the prognosis of IDA with IMH according to evaluation of neovascularisation using DCE-MRI.
With the application of DCE-MRI analysis, a theoretical basis for the prognosis of IDA with IMH may be provided for physicians. In the future, results from our study may help patients with such lesions receive an optimal and more reasonable treatment strategy, thus improving the prognosis of IDA with IMH in clinical practice.
References
Footnotes
Correction notice This paper has been updated since first published to update article type.
Contributors ZT performed the manuscript writing. YZ and XY made critical revision to the manuscript for important intellectual content. ZW, WL, WZ and JL all participated in the final design of the study. YZ conceived and designed the research, and handled funding and supervision. All authors read and approved the final manuscript.
Funding This work was supported by the National Natural Science Foundation of China (grant numbers: 81801158, 81801156, 81471167 and 81671139) and the Special Research Project for Capital Health Development (grant number: 2018-4-1077).
Competing interests None declared.
Patient consent for publication Not required.
Ethics approval The research was permitted by the ethics committee of Beijing Tian Tan Hospital (KY 2019-024-03) and written informed consents would be obtained from all patients included in this study. This study adheres to the ethical principles of the Declaration of Helsinki and Human Biomedical Research Ethical Issues and Policy Guidance.
Provenance and peer review Not commissioned; externally peer reviewed.
Data availability statement Data are available upon reasonable request.