Article Text
Abstract
Background The effect of cerebral small vessel disease (CSVD) and intracranial arterial stenosis (ICAS) on stroke outcomes remains unclear.
Methods Data of 1045 patients with minor stroke or transient ischaemic attack (TIA) were obtained from 45 sites of the Clopidogrel in High-Risk Patients with Acute Non-disabling Cerebrovascular Events (CHANCE) trial. We assessed the associations of burdens of CSVD and ICAS with new strokes and bleeding events using multivariate Cox regression models and those with modified Rankin Scale (mRS) scores using ordinal logistic regression models.
Results Among the 1045 patients, CSVD was present in 830 cases (79.4%) and ICAS in 460 (44.0%). Patients with >1 ICAS segment showed the highest risk of new strokes (HR 2.03, 95% CI 1.15 to 3.56, p=0.01). No association between CSVD and the occurrence of new strokes was found. The presence of severe CSVD (common OR (cOR) 2.01, 95% CI 1.40 to 2.89, p<0.001) and >1 ICAS segment (cOR 2.15, 95% CI 1.57 to 2.93, p<0.001) was associated with higher mRS scores. Severe CSVD (HR 10.70, 95% CI 1.16 to 99.04, p=0.04), but not ICAS, was associated with a higher risk of bleeding events. Six-point modified CSVD score improved the predictive power for bleeding events and disability.
Interpretation CSVD is associated with more disability and bleeding events, and ICAS is associated with an increased risk of stroke and disability in patients with minor stroke and TIA at 3 months. CSVD and ICAS may represent different vascular pathologies and play distinct roles in stroke outcomes.
Trial registration number NCT00979589
- cerebral small vessel disease
- intracranial arterial stenosis
- stroke
This is an open access article distributed in accordance with the Creative Commons Attribution Non Commercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited, appropriate credit is given, any changes made indicated, and the use is non-commercial. See: http://creativecommons.org/licenses/by-nc/4.0/.
Statistics from Altmetric.com
Introduction
Minor ischaemic stroke and transient ischaemic attack (TIA) are two common types of acute cerebrovascular disease.1 Studies have found that intracranial arterial stenosis (ICAS) is a crucial cause and pathology of ischaemic stroke, and increases the risk of stroke recurrence.2–4 Although it has been shown that cerebral small vessel disease (CSVD) increased the risk of incident stroke in the elderly population,5 whether CSVD is associated with stroke recurrence regarding secondary stroke prevention has been controversial. A post hoc analysis of the Stenting and Aggressive Medical Management for Preventing Recurrent Stroke in Intracranial Stenosis (SAMPPRIS) trial showed a lack of association between CSVD and stroke recurrence,6 while a study investigating the Oxford Vascular Study Cohort (OXVASC) and University of Hong Kong (HKU) cohort reported the opposite.7 Despite of previous meta-analysis indicating that CSVD may increase incident stroke in general population and in patients with stroke, publication bias have been noted.5 8 It has been highlighted that CSVD and ICAS may be two distinct entities, and vascular mechanism only accounts for no >2% of white matter hyperintensity (WMH) compared with 70% for large-artery atheromatous disease.9 With different mechanisms, CSVD and ICAS may serve as separate vascular pathologies and play different roles in outcomes of ischaemic stroke. The questions whether, in what aspect and at what level CSVD and ICAS are associated with the outcomes of ischaemic stroke remain unanswered.
CSVD burden score defined by MRI can capture the overall profile CSVD imaging markers, and it has been shown to be associated with cognitive impairment,10 lower quality of life11 and increased all-cause mortality after stroke.12 More recently, a 6-point modified version of CSVD burden score which stratified different severity of WMH and basal ganglia perivascular space (BG-PVS) has been proposed, showing better predictive value for intracranial haemorrhage in patients with TIA/ischaemic stroke.7 However, the validity of the 6-point modified CSVD burden score needs to be tested.
The Clopidogrel in High-Risk Patients with Acute Nondisabling Cerebrovascular Events (CHANCE) randomised clinical trial13 14 provides a valuable dataset to evaluate how CSVD and ICAS burdens, separately or by interaction, are associated with different outcomes of stroke. Therefore, in this prespecified imaging substudy of the CHANCE trial, we aim to investigate the roles of CSVD and ICAS in different outcomes (ie, new stroke, modified Rankin Scale (mRS) score and any bleeding event) at 3-month follow-up in patients with minor stroke and TIA, and to test the validity of the 6-point modified CSVD burden score for predicting poststroke outcomes.
Methods
Overview of the CHANCE trial and the imaging substudy
The CHANCE trial was a randomised, double-blind, placebo-controlled clinical trial conducted at 114 clinical centres in China from October 2009 to July 2012. Details of the design, rationale and major results have been previously described.13 14 Data of the present subgroup analysis were derived from the prespecified imaging substudy of the CHANCE trial.2 3 15 MRI examinations (3.0 or 1.5 T), including T1-weighted imaging, T2-weighted imaging, diffusion-weighted imaging and three-dimensional time-of-flight magnetic resonance angiography (MRA), were required in all substudy patients. Patients without a baseline MRI examination or with incomplete sequences to assess ICAS or CSVD were excluded from the present analysis. MRI images were obtained in 1045 patients for final interpretation and analysis in this study.
Image interpretation and evaluation
MRI data were collected from individual centres in digital format and were evaluated centrally in Beijing Tiantan Hospital. All imaging readers were blinded to each other and to patients’ information.
Imaging markers of CSVD were evaluated by two readers (LZ and YX) according to the STandards for ReportIng Vascular changes on nEuroimaging.16 In the calculation of the 4-point CSVD burden score, 1 point was allocated to each of the following MRI parameters7: severe WMH (periventricular WMH (PV-WMH) Fazekas grade 3 or deep WMH Fazekas grade 2–3), the presence of lacune, the presence of microbleeds and moderate-to-severe BG-PVS (n>10), yielding a score ranging from 0 to 4 points. Patients with CSVD were further categorised into those with slight CSVD (burden score 1–2) and those with severe CSVD (burden score 3–4).
Moreover, Lau et al proposed a 6-point modified CSVD burden score which may improve the predictive power of CSVD for intracranial haemorrhage in two stroke cohorts.7 Accordingly, we also calculated the modified CSVD burden score by allocating 1 point for the presence of lacunes, 1–4 microbleeds, >20 BG-PVS, moderate WMH (total PV-WMH+deep WMH Fazekas grade 3–4); and allocating 2 points for >5 microbleeds and severe WMH (total PV-WMH+deep WMH Fazekas grade 5–6).7 Patients were also categorised into those with slight CSVD (modified burden score 1–2) and those with severe CSVD (modified burden score 3–6).
ICAS was evaluated by two other readers (XZ and JJ). An additional third reader (XM) resolved the disagreements >10%. The presence of ICAS was defined as 50%–99% stenosis according to the Warfarin-Aspirin Symptomatic Intracranial Disease (WASID) trial criteria17 or as an occlusion of the following arterial segments on MRA: intracranial segment of internal carotid arteries, middle cerebral arteries (M1/M2), intracranial segment of vertebral arteries and basilar artery. The severity of ICAS was classified into three levels according to the number of ICAS segments, that is, no ICAS, 1 ICAS segment and >1 ICAS segment. According to the combined features of CSVD and ICAS burdens, patients were further stratified into nine groups.
Efficacy and safety outcomes
In consistence with CHANCE trial, the primary efficacy outcome was a new stroke occurrence (ischaemic or haemorrhagic) at the 3-month follow-up. As no haemorrhagic stroke occurred in the present study, the primary efficacy outcome was, thus, equivalent to a new ischaemic stroke. The secondary efficacy outcome was the mRS score at 3 months. The safety outcome was any bleeding event during the 3-month follow-up.
Statistical analysis
Categorical variables were presented as percentages and continuous variables as mean with SD or median with IQR. Comparison of baseline variables among different CSVD and ICAS statuses were performed using χ2 test for categorical variables and one-way analysis of variance or Kruskal-Wallis test for continuous variables.
We present the time to the primary efficacy outcome event for each imaging group by Kaplan-Meier curves adjusted for age, gender, premorbid mRS score, antiplatelet therapy, body mass index, history of ischaemic stroke, TIA, myocardial infarction, congestive heart failure, known atrial fibrillation or flutter, hypertension, diabetes, hypercholesterolaemia, smoking status, time to randomisation, qualifying event and the National Institutes of Health Stroke Scale (NIHSS) score on admission. We assessed the associations of burdens of CSVD measured by 4-point and 6-point burden scores and ICAS with new stroke and bleeding events using multivariate Cox regression models and those with mRS scores using ordinal logistic regression models, first in the whole cohort and then in patients without new stroke occurrence (sensitivity analysis). Adjusted HRs or common ORs (cORs) and their 95% CIs were calculated based on two models. In the first model, we adjusted age, gender, premorbid mRS score and antiplatelet therapy. In the second model, we included all potential covariates (age, gender, premorbid mRS score, antiplatelet therapy, body mass index, history of ischaemic stroke, TIA, myocardial infarction, congestive heart failure, known atrial fibrillation or flutter, hypertension, diabetes, hypercholesterolaemia, smoking status, time to randomisation, qualifying event and NIHSS score on admission). We tested the statistical significance of CSVD×ICAS, CSVD×study treatment assignment, and ICAS×study treatment assignment in a multivariate Cox/ordinal logistic regression model to examine their interaction effect. The associations of each CSVD marker (BG-PVS, PV-WMH, deep WMH, lacune and microbleeds) with new stroke, bleeding events and mRS score were further assessed in multivariate Cox regression models and ordinal logistic regression models after adjusting for potential covariates. The level of significance was p<0.05 (two-sided). All analyses were performed with SAS V.9.4 (SAS Institute, Cary, North Carolina, USA). Data are available to researchers for reproducing the results or replicating the procedures by contacting the corresponding author.
Results
Patient demographics and baseline characteristics
A total of 1045 patients were included in the present analysis. Patients included in and excluded from the present substudy were balanced in most baseline variables (table 1). Demographic profile and clinical information of all included patients are shown in table 2. Patients with more severe CSVD or ICAS tended to be older in age, to have more history of ischaemic stroke, diabetes and hypertension, a longer time to randomisation, more strokes than TIAs as the qualifying event, a higher NIHSS score on admission and a higher occurrence of a new stroke at 3-month follow-up.
Comparison in demographic profiles between the imaging substudy population and the excluded population
Baseline characteristics of patients included in the present substudy of CHANCE trial
Association between CSVD/ICAS status and new stroke
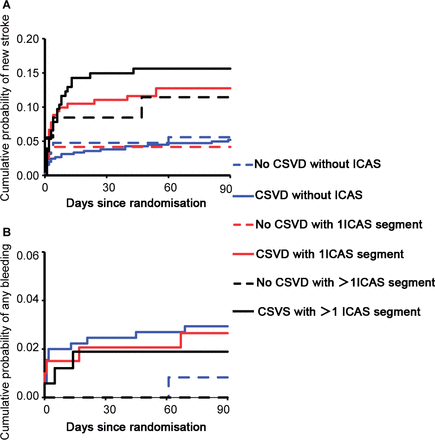
During the 3-month follow-up, new strokes occurred in 13 patients (6.0%) without CSVD, 51 (10.5%) with slight CSVD and 22 (6.4%) with severe CSVD and in 31 patients (5.3%) without ICAS, 26 (10.4%) with 1 ICAS segment and 29 (13.8%) with >1 ICAS segment. The Kaplan-Meier curves showed that patients presenting with ICAS displayed a higher occurrence of a new stroke at 3 months than those without (table 3; figure 1A; log-rank p=0.001). The presence of ICAS, but not CSVD at any level measured by 4-point or 6-point burden scores, was significantly associated with an increased 3-month risk of a new stroke (for 1 ICAS segment, HR 1.75, 95% CI 1.01 to 3.05, p=0.047; for >1 ICAS segment, HR 2.03, 95% CI 1.15 to 3.56, p=0.01) after adjusting for all potential covariates (table 4). No interaction was observed between CSVD and ICAS, CSVD and treatment assignment and between ICAS and treatment assignment on the primary efficacy outcome (p for interaction=0.51, 0.87 and 0.36 for a test across all the groups in the fully adjusted model, respectively). Furthermore, no CSVD marker was significantly associated with new stroke occurrence at 3 months (table 5).
Cumulative probability of new stroke and any bleeding event by CSVD and ICAS status. (A) Kaplan-Meier curves showing more occurrences of new strokes in patients with ICAS in a dosage-dependent way; (B) Kaplan-Meier curves showing that more bleeding events occurred in patients with CSVD than in those without. The solid black line overlapped with dashed red line, and the latter is hidden. CSVD, cerebral small vessel disease; ICAS, intracranial arterial stenosis.
The association between CSVD/ICAS status and the 3-month new stroke and bleeding events
Multivariate regression for the association between CSVD or ICAS burdens and the 3-month prognosis in the entire cohort
Association between CSVD imaging markers and new stroke at 3 months
Association between CSVD/ICAS status and mRS
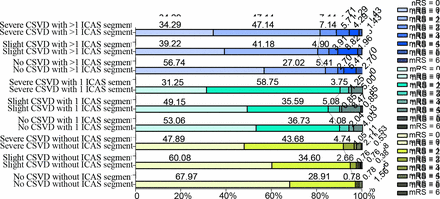
A higher proportion of disability at 3 months was observed among patients with CSVD and ICAS (figure 2). CSVD (for slight CSVD, cOR 1.45, 95% CI 1.04 to 2.03, p=0.03; for severe CSVD, cOR 2.01, 95% CI 1.40 to 2.89, p<0.001) and ICAS (for 1 ICAS segment, cOR 1.73, 95% CI 1.29 to 2.32, p<0.001; for >1 ICAS segment, cOR 2.15, 95% CI 1.57 to 2.93, p<0.001) were both associated with a higher mRS scores (indicating a poorer functional outcome) at 3 months in the first model. After further adjustment for other potential covariates, the association between severe CSVD and mRS score did not reach the significance level but still showed the same tendency (cOR 1.46, 95% CI 0.98 to 2.17, p=0.06). Using the 6-point modified CSVD burden score, CSVD showed stronger association with 3-month disability. Defined by the modified score, both slight CSVD (cOR 1.53, 95% CI 1.13 to 2.08, p=0.007) and severe CSVD (cOR 2.15, 95% CI 1.45 to 3.21, p<0.001) were associated with a higher mRS scores at 3 months in the first model. After further adjustment for other potential covariates, the association remained significant in our sensitivity analysis (for slight CSVD, cOR 1.42, 95% CI 1.02 to 1.98, p=0.04; for severe CSVD, cOR 1.55, 95% CI 1.00 to 2.40, p=0.05) (table 4).
Modified Rankin Scale (mRS) distributions at 3 months in different CSVD and ICAS statuses. Both higher levels of CSVD severity and more ICAS segments caused a more severe disability among the groups. CSVD, cerebral small vessel disease; ICAS, intracranial arterial stenosis.
No interaction effect between CSVD and ICAS was observed (p for interaction=0.97 in the fully adjusted model). The main finding was preserved in the multivariate regression analysis in sensitivity analysis for patients without new stroke occurrence (n=959), where severe CSVD (cOR 2.20, 95% CI 1.49 to 3.24, p<0.001) and ICAS (for 1 ICAS segment, cOR1.55, 95% CI 1.13 to 2.13, p=0.007; for >1 ICAS segment, cOR1.68, 95% CI 1.19 to 2.36, p=0.003) were both associated with a higher mRS scores at 3 months in the first model. In model 2, severe CSVD (cOR 1.61, 95% CI 1.05 to 2.48, p=0.03) and ICAS (for 1 ICAS segment, cOR1.64, 95% CI 1.16 to 2.32, p=0.005; for >1 ICAS segment, cOR1.56, 95% CI 1.07 to 2.26, p=0.02) still showed significant association with higher mRS scores.
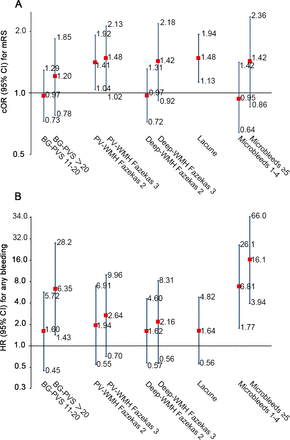
Among CSVD markers, severe PV-WMH (for PV-WMH Fazekas grade 2, cOR 1.41, 95% CI 1.04 to 1.92; for PV-WMH Fazekas grade 3, cOR 1.48, 95% CI 1.02 to 2.13) and the presence of lacune (cOR 1.48, 95% CI 1.13 to 1.94) were associated with a higher disability score (figure 3A).
Association between CSVD imaging markers and mRS scores or bleeding events at 3 months. (A) The parameters PV-WMH and lacune were predictive of a poor functional outcome; (B) Microbleeds and advanced BG-PVS (>20) were suggestive of more bleeding events at 3 months. BG-PVS, basal ganglia perivascular space; cOR, common OR; CSVD, cerebral small vessel disease; mRS, modified Rankin Scale; PV-WMH, periventricular white matter hyperintensity.
Association between CSVD/ICAS status and bleeding events
A bleeding event occurred in 23 (2.2%) patients in the substudy during the 3-month follow-up. No intracranial bleeding was observed in any group. The Kaplan-Meier curves further showed that the prevalence of a bleeding event during follow-up was higher in patients with CSVD than in those without (table 3; figure 1B; log-rank p=0.502). Severe CSVD, rather than ICAS, was associated with a bleeding event in the multivariate regression models (in model 1, HR 8.21, 95% CI 1.00 to 67.48, p=0.05; in model 2, HR 10.70, 95% CI 1.16 to 99.04, p=0.04). Analysis using the 6-point modified version of CSVD burden score showed stronger association between CSVD and 3-month bleeding events (in model 1, HR 12.44, 95% CI 1.36 to 115.46, p=0.03; in model 2, HR 48.65, 95% CI 2.10 to 1124.88, p=0.02 for severe CSVD) (table 4).
No interaction was observed between CSVD and ICAS, CSVD and treatment assignment or ICAS and treatment assignment on the safety outcome (p for interaction=0.65, 0.99 and 0.82 in the fully adjusted model, respectively). CSVD was still marginal associated with an increased risk of bleeding in the subgroup of patients without new stroke occurrence (HR 9.60, 95% CI 0.99 to 92.99, p=0.051). Among CSVD markers, BG-PVS >20 (HR 6.35, 95% CI 1.43 to 28.2) and the presence of microbleeds (for microbleeds 1–4, HR 6.81, 95% CI 1.77 to 26.1; for microbleeds >5, HR 16.1, 95% CI 3.94 to 66.0) were associated with an increased risk of a bleeding event at 3 months (figure 3B).
Discussion
In this substudy of the CHANCE trial, we investigated how CSVD and ICAS were associated with new stroke, mRS score and bleeding events at 3-month follow-up in patients with minor stroke and TIA. CSVD was found to be associated with the parameters mRS score and bleeding events, but not new stroke at 3 months. By contrast, ICAS was shown to be closely associated with new stroke and mRS score, rather than bleeding events. The 6-point modified version of CSVD burden score showed stronger association with bleeding events and disability than the 4-point version. This result implies that CSVD and ICAS contribute differently to stroke outcomes and both deserve heightened clinical attention in the secondary prevention of minor stroke and TIA. The 6-point modified version of CSVD burden score may surpass the 4-point version in predicting bleeding events and disability for minor stroke and TIA.
ICAS is a crucial aetiology of stroke and plays a significant role in the prognosis. In minor stroke and TIA, the presence of severe ICAS or occlusion doubles the risk of recurrent stroke.3 Moreover, ≥70% ICAS doubles the risk of stroke recurrence in the stenotic territory compared with 50%–69% stenosis.18 Our findings further suggest that the number of ICAS segments, symptomatic or not, may be associated with stroke recurrence in a dosage-dependent way.
Unlike ICAS, however, CSVD was not significantly associated with new strokes at 3 months. Our finding is supported by a post hoc analysis of the SAMPPRIS study.6 However, an analysis comprising OXVASC and HKU cohorts reported the opposite.7 Potential explanations are outlined as follows. First, different study design and patient selection may be one reason. Second, Lau et al did not rule out other potentially confounding factors especially the ICAS status in their analysis, while CSVD and ICAS share some common risk factors and tend to coexist.6 19 Although previous meta-analysis reported significant association between CSVD and stroke in both general population and stroke patient, publication bias has also been revealed.5 8 These results may suggest the CSVD may not exert significant influence over the secondary stroke prevention. The role of CSVD in stroke recurrence will need more investigations.
Moreover, our findings indicate that CSVD, especially the parameters PV-WMH and lacune, are associated with a 3-month functional decline of ischaemic cerebral events, independent of premorbid mRS score. The different contributions of PV-WMH and deep WMH suggest that they may differ in their underlying mechanisms, in which a non-ischaemic origin may result in PV-WMH.20 Up to now, the association between CSVD and functional outcome in the context of acute ischaemic stroke has only been demonstrated in several small-sample studies.21 22 Given that CSVD mainly affects the daily function domains,19 23 it seems reasonable to observe that the presence of CSVD is predictive of a higher mRS score.
Our finding that CSVD especially microbleeds was associated with bleeding events independent of common risk factors suggests that intracranial and extracranial haemorrhagic events may be related and denote a systemic arteriolar dysfunction. Although increased CSVD burden may increase the risk of intracranial haemorrhage in patients with ischaemic stroke,24 25 this association was not investigated in the CHANCE population as no intracranial haemorrhage was observed in this substudy. Likewise, enlarged PVSs in the basal ganglia may also be pathological, especially when their number exceeds 20 in one hemisphere. However, the post hoc analytic approach may result in false-positive findings, and the mechanism underlying the association between microbleeds or PVS and bleeding events needs further investigation. Previous meta-analysis suggested that ICAS may be associated with microbleeds,26 our investigation did not find significant interaction between CSVD burden and ICAS, while CSVD may be associated with haemorrhagic events independently.
The use of CSVD burden score which integrates CSVD markers instead of a single marker has several strengths. The burden score can capture the overall severity of CSVD, and the total burden score may be more representative of CSVD as patients present with multiple imaging markers may be more likely to be associated with small vessel pathology than patients with mere WMH or lacune. Moreover, it may provide a simple way to evaluate CSVD in practice. The 4-point CSVD burden score has been widely used, and has been shown to be associated with cognitive impairment,10 lower quality of life11 and increased all-cause mortality after stroke.12 More recently, the 6-point modified CSVD burden which stratified different severity of WMH and BG-PVS has been proposed, showing better predictive value for intracranial haemorrhage in patients with TIA/ischaemic stroke.7 Our result also demonstrates that the modified version of CSVD burden score may outperform the 4-point version in predicting bleeding events and disability after minor stroke and TIA.
Our findings have several implications. First, our results indicate that the ICAS increased risk in stroke recurrence and disability in minor stroke and TIA. Second, attention should as well be paid to CSVD in ischaemic stroke to decrease the severity of disability, despite that it may not increase the risk of stroke recurrence. Third, ICAS largely represents atherosclerosis due to vascular risk factors, while inflammation processes, blood-brain barrier dysfunctions and impaired vascular regulation may underlie the mechanisms responsible for CSVD. Further studies investigating the contribution of CSVD-related pathophysiology to stroke outcomes are needed. Fourth, we suggest that future studies should take ICAS feature into consideration when exploring the consequence of CSVD imaging markers. Fifth, 6-point modified version of CSVD burden score, compared with 4-point version, may have better predictive value for functional outcome and bleeding events in patients with minor stroke and TIA.
The present study has several limitations. First, this study involved less than half of all CHANCE participating sites. The included individuals tended to be older in age and experienced more ischaemic stroke than the whole CHANCE population, which could potentially confound results. However, the current data were derived from prespecified centres, and the current population showed balanced characteristics in most demographic variables compared with the excluded CHANCE population. Second, all patients were Chinese in the CHANCE trial, which may limit the generalisability of the findings to other populations. Third, distal and <50% ICAS, as well as anterior and posterior arteries were not measured, which may underestimate the ICAS burden in patients. However, the distal arterial stenosis is not accurately detected by MR angiography, and our ICAS definition was consistent with WASID criteria.17 Fifth, mRS was used as a general measurement of functional domain, which did not provide detailed information on patients’ daily function s such as gait, cognition and mood. Future studies delving into the influence of CSVD on detailed and multidomain functions after stroke are needed. Sixth, post hoc analysis and multiple hypothesis testing may lead to false-positive findings. More studies are still needed to confirm the role of CSVD with our without ICAS in stroke prognosis.
In conclusion, the presence of CSVD may be associated with more disability and bleeding events, while ICAS may be associated with an increased risk of new strokes and disability at 3 months in patients with minor stroke and TIA. The presence of CSVD and ICAS may represent distinct vascular pathologies and affect different aspects of stroke outcomes. The 6-point modified version of CSVD burden score may have better predictive value for stroke outcomes than the 4-point version.
References
Footnotes
Contributors YiW had full access to all of the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis. Study concept and design: HC, YP, YiW, YoW. Acquisition, analysis or interpretation of data: HC, YP, HY, JJ, XZ, LZ, YX, YiW. Drafting of the manuscript: HC. Critical revision of the manuscript and important intellectual contribution: SCJ, YiW, YoW. Statistical analysis: HY, HL. Obtained funding: YiW, YoW. Administrative, technical or material support: XZ, LL, JJ, YW. Study supervision: SCJ, YoW.
Funding This study is supported by grants from the Ministry of Science and Technology of the People’s Republic of China (2016YFC0901001, 2016YFC0901002, 2017YFC1307900, 2017YFC1310901, 2018YFC1311700 and 2018YFC1311706), Beijing Municipal Committee of Science and Technology (D151100002015003, Beijing Excellent Talents Training and Supporting Top Youth Team, D171100003017001 and 2016000021223TD03), Beijing Municipal Commission of Health and Family Planning (No.2016-1-2041, SML20150502), National Natural Science Foundation of China (81825007), Beijing Municipal Education Commission (Excellent Young Scientists Project) and the third batch of National Ten Thousand Talents Plan, and Beijing Outstanding Young Scientist Program (BJJWZYJH01201910025030).
Competing interests CJ is the principal investigator of the POINT trial, a NIH-sponsored trial with clopidogrel and placebo donated by Sanofi.
Patient consent for publication Not required.
Ethics approval This study was approved by the ethics committee of Beijing Tiantan Hospital.
Provenance and peer review Not commissioned; internally peer reviewed.
Data availability statement Data are available on reasonable request.